668478
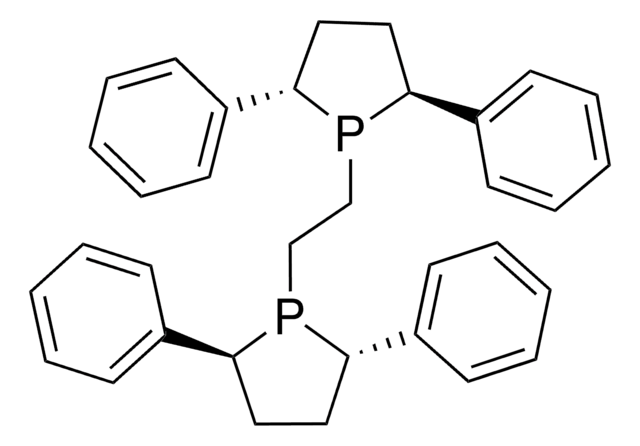
(+)-1,2-Bis[(2R,5R)-2,5-diethylphospholano]ethane
kanata purity
Synonym(s):
(R,R)-Et-BPE
About This Item
Recommended Products
Quality Level
refractive index
n20/D 1.5249
bp
104-106 °C/0.05 mmHg
density
0.939 g/mL at 25 °C
functional group
phosphine
SMILES string
CC[C@@H]1CC[C@@H](CC)P1CCP2[C@H](CC)CC[C@H]2CC
InChI
1S/C18H36P2/c1-5-15-9-10-16(6-2)19(15)13-14-20-17(7-3)11-12-18(20)8-4/h15-18H,5-14H2,1-4H3/t15-,16-,17-,18-/m1/s1
InChI key
QOLRLVPABLMMKI-BRSBDYLESA-N
Application
- Chromium diphosphine chloride complex which is employed as a catalyst for chemoselective oligomerization reaction.
- α-Arylpyrrolidines by Suzuki-Miyaura cross-coupling and enantioselective copper-catalyzed intramolecular hydroamination reactions.
It can also be used as a catalyst in the enantioselective preparation of (perfluoroalkyl)butenyldiketones via cross Rauhut-Currier reaction of β-perfluoroalkylenones and vinyl ketones.
Legal Information
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
Asymmetric hydrogenation reactions represent the ideal process for the commercial manufacture of single-enantiomer compounds, because of the ease by which these robust procedures can be scaled up and because of the low levels of byproducts generated in these asymmetric hydrogenations.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![(+)-1,2-Bis[(2S,5S)-2,5-dimethylphospholano]benzene kanata purity](/deepweb/assets/sigmaaldrich/product/structures/319/912/cec7b70f-bf7c-4a96-9f11-a73ae892e34c/640/cec7b70f-bf7c-4a96-9f11-a73ae892e34c.png)


![1,2-Bis[(2S,5S)-2,5-diisopropylphospholano]ethane kanata purity](/deepweb/assets/sigmaaldrich/product/structures/963/936/9ad14a4d-ae8f-4116-b620-9d86321bd1cb/640/9ad14a4d-ae8f-4116-b620-9d86321bd1cb.png)

![9-Borabicyclo[3.3.1]nonane solution 0.5 M in THF](/deepweb/assets/sigmaaldrich/product/structures/180/891/8b64e597-269d-4780-98b6-40889dfd06b9/640/8b64e597-269d-4780-98b6-40889dfd06b9.png)
