109428
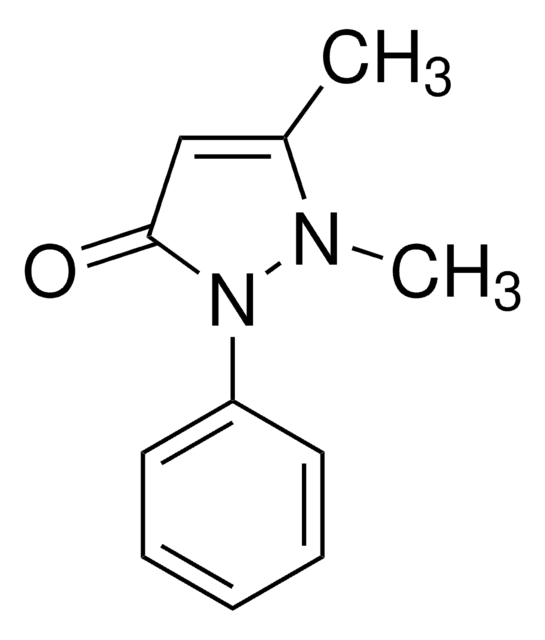
4-Hydroxyantipyrine
99%
Synonym(s):
4-Hydroxy-2,3-dimethyl-1-phenyl-3-pyrazolin-5-one, NSC 174055
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C11H12N2O2
CAS Number:
Molecular Weight:
204.23
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
99%
mp
184-186 °C (lit.)
functional group
ketone
SMILES string
CN1N(c2ccccc2)C(=O)C(O)=C1C
InChI
1S/C11H12N2O2/c1-8-10(14)11(15)13(12(8)2)9-6-4-3-5-7-9/h3-7,14H,1-2H3
InChI key
SKVPTPMWXJSBTF-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
4-Hydroxyantipyrine is formed during oxidative deamination of aminopyrine. It is a metabolite of antipyrine.
Application
4-Hydroxyantipyrine was used to study the relationships between the metabolism of antipyrine, hexobarbitone and theophylline in man. It was used in a study on flow injection analysis system for the characterisation of pharmaceutical compounds via combination of diode array UV, 1H NMR, FT-IR spectroscopy and time-of-flight mass spectrometry.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
M Monshouwer et al.
Xenobiotica; the fate of foreign compounds in biological systems, 25(5), 491-499 (1995-05-01)
1. In order to investigate the effect of a bacterial acute phase response model on drug disposition in vivo, plasma clearances of antipyrine, caffeine, paracetamol and indocyanine green were investigated in the healthy and Actinobacillus pleuropneumoniae-infected pig. 2. Indocyanine green
G Engel et al.
Clinical pharmacology and therapeutics, 59(6), 613-623 (1996-06-01)
Antipyrine has been widely used as a probe drug for human oxidative drug metabolism. To evaluate the role of antipyrine as a model drug, we have identified the cytochrome P450 enzymes involved in 4-hydroxyantipyrine, 3-hydroxymethylantipyrine, and norantipyrine formation. We used
Reaction of drugs with nitrous acid as a source of carcinogenic nitrosamines.
W Lijinsky
Cancer research, 34(1), 255-258 (1974-01-01)
S B Seredenin et al.
Biulleten' eksperimental'noi biologii i meditsiny, 110(11), 491-493 (1990-11-01)
Antipyrine oxidation was studied in C57BL/6 and BALB/c inbred mice. It was found that C57BL/6 are weak oxidant but BALB/c are strong oxidants of antipyrine. Animals F1 hybrids inherited the high capacity of antipyrine oxidation.
R P Shrewsbury et al.
Research communications in chemical pathology and pharmacology, 64(3), 455-462 (1989-06-01)
Antipyrine metabolism was determined after hemodilution with 40 ml/kg of Fluosol in conscious, unrestrained female and male rats. Rats received an intravenous antipyrine dose (20 mg/kg) 24, 48, or 72 hours after hemodilution and the pharmacokinetic parameters were compared to
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service