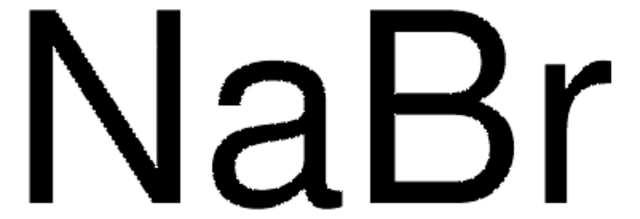Alle Fotos(4)
Wichtige Dokumente
P1147
Kaliumacetat
ReagentPlus®, ≥99.0%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(4)
About This Item
Lineare Formel:
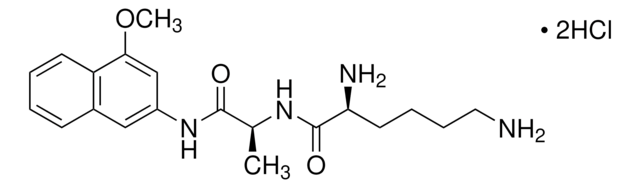
CH3COOK
CAS-Nummer:
Molekulargewicht:
98.14
Beilstein:
3595449
EG-Nummer:
MDL-Nummer:
UNSPSC-Code:
12352302
PubChem Substanz-ID:
NACRES:
NA.21
Empfohlene Produkte
Qualität
reagent
Dampfdruck
<0.0000001 hPa ( 25 °C)
Produktlinie
ReagentPlus®
Assay
≥99.0%
Form
powder or crystals
pH-Wert
7-9 (25 °C, 98.2 g/L)
Dichte
1.57 g/cm3 at 25 °C (lit.)
SMILES String
[K+].CC([O-])=O
InChI
1S/C2H4O2.K/c1-2(3)4;/h1H3,(H,3,4);/q;+1/p-1
InChIKey
SCVFZCLFOSHCOH-UHFFFAOYSA-M
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Allgemeine Beschreibung
Potassium acetate is an osmotic agent that can be prepared by the reaction between acetic acid and potassium hydroxide. It has replaced urea and glycol for deicing process. Its potential as a substitute to calcium chloride for osmotic distillation has been investigated. It intercalates with the hydroxyl groups on the kaolinite surface leading to surface modification. It acts as a catalyst that accelerates the acetylation of wood at low temperature.
Anwendung
Potassium acetate has been used during granule bound starch synthase (GBSS) activity assay. It may be used as an activator to produce waste tea activated carbon (WTAC).
Rechtliche Hinweise
ReagentPlus is a registered trademark of Merck KGaA, Darmstadt, Germany
Lagerklassenschlüssel
13 - Non Combustible Solids
WGK
WGK 1
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Potassium acetate-catalyzed acetylation of wood: reaction rates at low temperatures.
Obataya E and Minato K.
Wood Science and Technology, 43(5-6), 405-413 (2009)
M M Shah et al.
Applied biochemistry and biotechnology, 63-65, 423-433 (1997-04-01)
Potassium acetate is currently made by reacting petroleum-based acetic acid with potassium hydroxide. An alternate process, anaerobic fermentation of dextrose with Clostridium thermoaceticum, could be used and could possibly be cheaper. Growth characteristics and productivity of the fermentation were optimized
Purification of starch granules from Arabidopsis leaves and determination of granule-bound starch synthase activity.
Albi T, et al.
Bio-protocol, 4(23), e1316-e1316 (2013)
Preparation of waste tea activated carbon using potassium acetate as an activating agent for adsorption of Acid Blue 25 dye.
Auta M and Hameed BH.
Chemical Engineering Journal, 171(2), 502-509 (2011)
Modification of kaolinite surfaces through intercalation with potassium acetate, II.
Frost RL, et al.
Journal of Colloid and Interface Science, 214(1), 109-117 (1999)
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.