76547
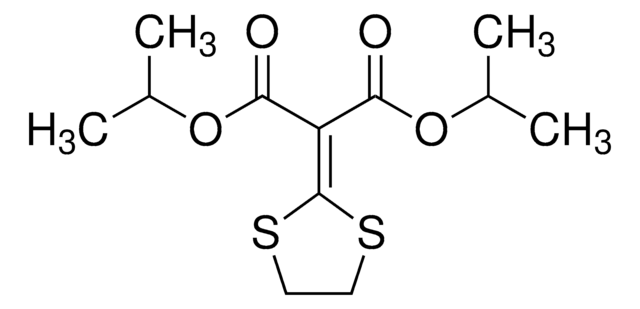
Isoprothiolan
PESTANAL®, analytical standard
Synonym(e):
2-(1,3-Dithiolan-2-yliden)-malonsäure-diisopropylester
About This Item
Empfohlene Produkte
Qualität
analytical standard
Qualitätsniveau
Produktlinie
PESTANAL®
Assay
≥98.0% (HPLC)
Haltbarkeit
limited shelf life, expiry date on the label
Methode(n)
HPLC: suitable
gas chromatography (GC): suitable
Anwendung(en)
agriculture
environmental
Format
neat
Lagertemp.
2-8°C
SMILES String
CC(C)OC(=O)\C(C(=O)OC(C)C)=C1/SCCS1
InChI
1S/C12H18O4S2/c1-7(2)15-10(13)9(11(14)16-8(3)4)12-17-5-6-18-12/h7-8H,5-6H2,1-4H3
InChIKey
UFHLMYOGRXOCSL-UHFFFAOYSA-N
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Allgemeine Beschreibung
Anwendung
Verpackung
Rechtliche Hinweise
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.