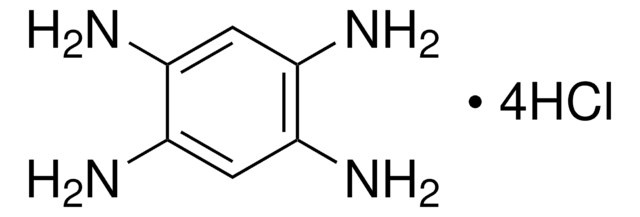
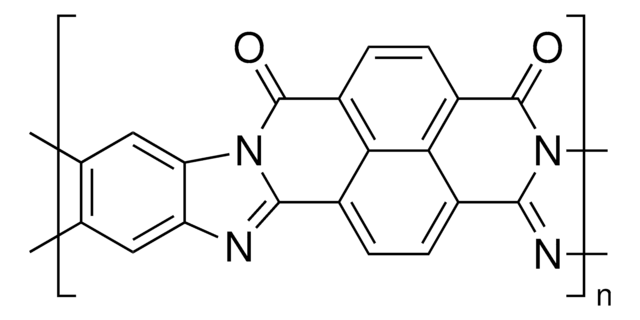
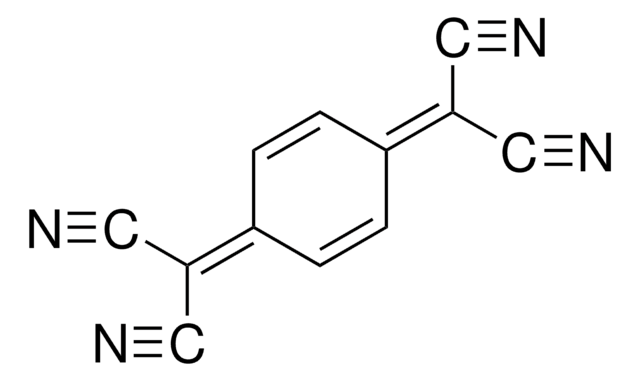
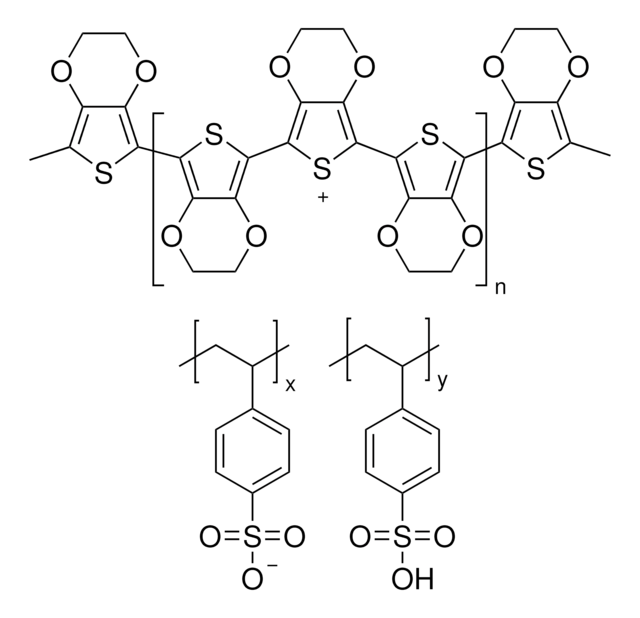
776734
4-(2,3-Dihydro-1,3-dimethyl-1H-benzimidazol-2-yl)-N,N-dimethylbenzenamine
97% (HPLC)
Synonym(e):
4-(1,3-Dimethyl-2,3-dihydro-1H-benzoimidazol-2-yl)phenyl)dimethylamine, N-DMBI
About This Item
Empfohlene Produkte
Qualitätsniveau
Assay
97% (HPLC)
Form
solid
mp (Schmelzpunkt)
105-110 °C
SMILES String
CN(C)c1ccc(cc1)C2N(C)c3ccccc3N2C
InChI
1S/C17H21N3/c1-18(2)14-11-9-13(10-12-14)17-19(3)15-7-5-6-8-16(15)20(17)4/h5-12,17H,1-4H3
InChIKey
AKIIMLCQTGCWQQ-UHFFFAOYSA-N
Allgemeine Beschreibung
Anwendung
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Acute Tox. 4 Oral
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Artikel
Solution-processed organic photovoltaic devices (OPVs) have emerged as a promising clean energy generating technology due to their ease of fabrication, potential to enable low-cost manufacturing via printing or coating techniques, and ability to be incorporated onto light weight, flexible substrates.
Fabrication procedure of organic field effect transistor device using a soluble pentacene precursor.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.
![Poly-[bis-(4-phenyl)-(2,4,6-trimethylphenyl)-amin] a poly(triaryl amine) semiconductor](/deepweb/assets/sigmaaldrich/product/structures/122/933/c34a34ab-284f-4890-adb8-126247a91d9b/640/c34a34ab-284f-4890-adb8-126247a91d9b.png)