D1446
Dexrazoxane
≥95% (HPLC)
Sinónimos:
(+)-(S)-4,4′-Propylenedi-2,6-piperazinedione, (S)-(+)-1,2-Bis(3,5-dioxopiperazin-1-yl)propane, Cardioxane, ICRF-187, NSC169780, Zinecard
About This Item
Productos recomendados
Quality Level
assay
≥95% (HPLC)
form
powder
color
white to off-white
solubility
DMSO: >20 mg/mL
originator
Johnson & Johnson
storage temp.
room temp
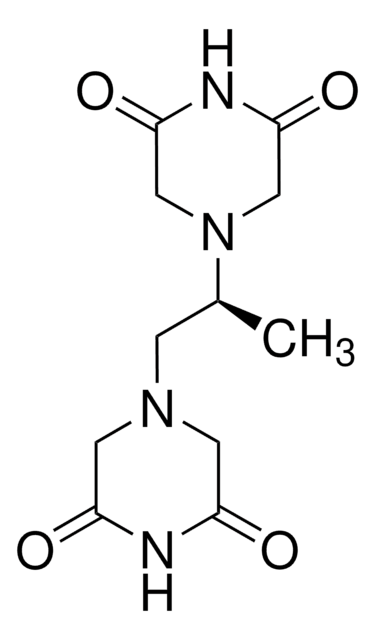
SMILES string
C[C@@H](CN1CC(=O)NC(=O)C1)N2CC(=O)NC(=O)C2
InChI
1S/C11H16N4O4/c1-7(15-5-10(18)13-11(19)6-15)2-14-3-8(16)12-9(17)4-14/h7H,2-6H2,1H3,(H,12,16,17)(H,13,18,19)/t7-/m0/s1
InChI key
BMKDZUISNHGIBY-ZETCQYMHSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
Application
Biochem/physiol Actions
Features and Benefits
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Contenido relacionado
n proliferating cells, the cell cycle consists of four phases. Gap 1 (G1) is the interval between mitosis and DNA replication that is characterized by cell growth. Replication of DNA occurs during the synthesis (S) phase, which is followed by a second gap phase (G2) during which growth and preparation for cell division occurs. Together, these three stages comprise the interphase phase of the cell cycle. Interphase is followed by the mitotic (M) phase.
Apoptosis, or programmed cell death (PCD), is a selective process for the removal of unnecessary, infected or transformed cells in various biological systems. As it plays a role in the homeostasis of multicellular organisms, apoptosis is tightly regulated through two principal pathways by a number of regulatory and effector molecules.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico