47633
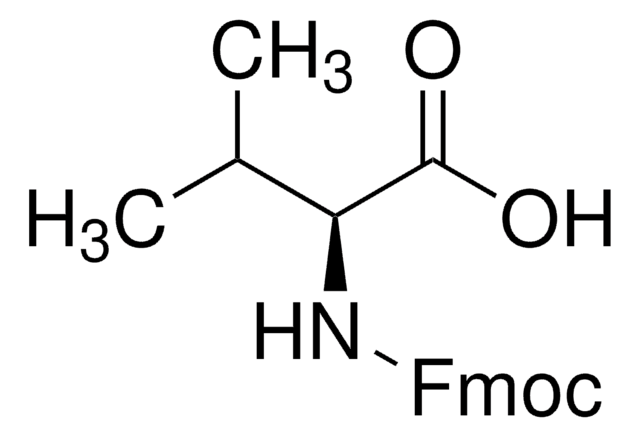
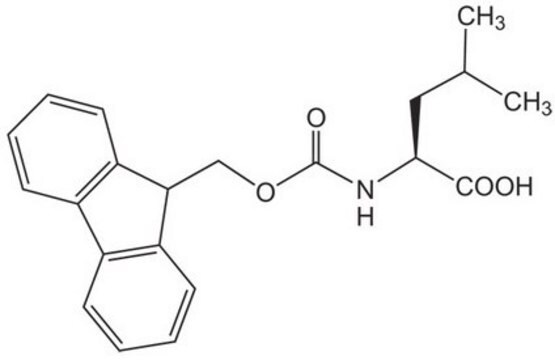
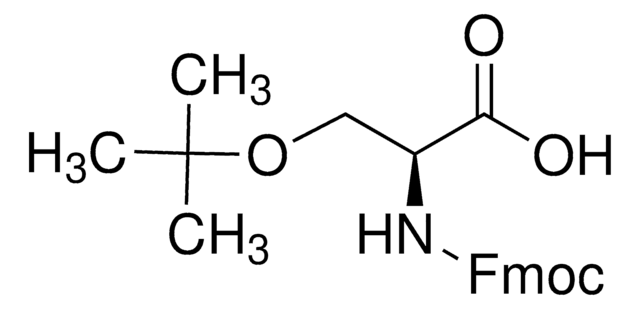
Fmoc-Leu-OH
≥97.0%
Sinónimos:
N-(9-Fluorenylmethoxycarbonyl)-L-leucine, Fmoc-L-leucine
About This Item
Productos recomendados
Quality Level
assay
≥97.0%
optical activity
[α]20/D −25±2°, c = 1% in DMF
reaction suitability
reaction type: C-H Activation
reaction type: Fmoc solid-phase peptide synthesis
reagent type: ligand
reaction type: Peptide Synthesis
mp
152-156 °C (lit.)
152-156 °C
application(s)
peptide synthesis
functional group
Fmoc
amine
carboxylic acid
storage temp.
2-8°C
SMILES string
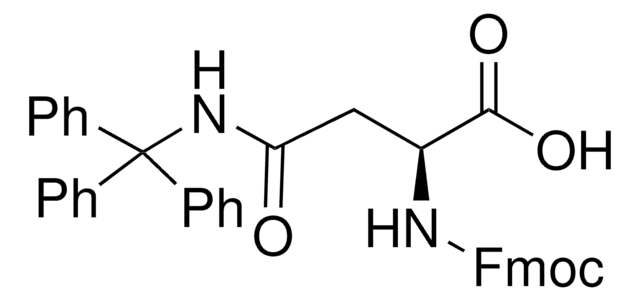
CC(C)C[C@H](NC(=O)OCC1c2ccccc2-c3ccccc13)C(O)=O
InChI
1S/C21H23NO4/c1-13(2)11-19(20(23)24)22-21(25)26-12-18-16-9-5-3-7-14(16)15-8-4-6-10-17(15)18/h3-10,13,18-19H,11-12H2,1-2H3,(H,22,25)(H,23,24)/t19-/m0/s1
InChI key
CBPJQFCAFFNICX-IBGZPJMESA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Application
- Various oligopeptides by reacting with functionalized α-amino acid hydrochloride salts.
- A cyclic depsipeptide sansalvamide A, a natural product found in marine fungus.
- Streptocidin A−D, decapeptide antibiotics naturally found in Streptomyces sp. Tü 6071.
- Coumaroyl dipeptide amide that can be used for cosmetic applications.
Biochem/physiol Actions
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
With a growing peptide drug market the fast, reliable and uncomplicated synthesis of peptides is of paramount importance.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico