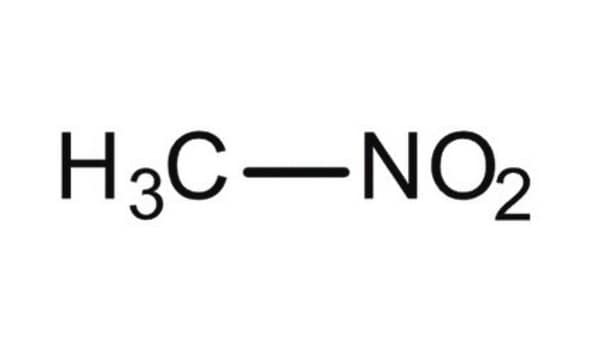
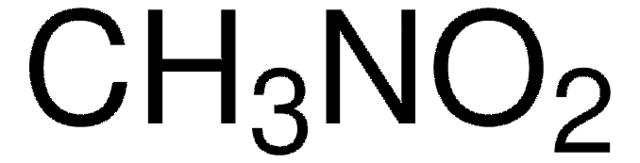About This Item
Recommended Products
vapor density
2.1 (vs air)
Quality Level
vapor pressure
2.7 mmHg
Assay
≥96%
form
liquid
autoignition temp.
784 °F
expl. lim.
7.3 %, 33 °F
technique(s)
HPLC: suitable
impurities
<0.030% water
refractive index
n20/D 1.382 (lit.)
pH
6.4 (20 °C, 0.01 g/L)
bp
101.2 °C (lit.)
mp
−29 °C (lit.)
density
1.127 g/mL at 25 °C (lit.)
λ
H2O reference
UV absorption
λ: 380 nm Amax: 1.00
λ: 386 nm Amax: 0.50
λ: 395 nm Amax: 0.20
λ: 400 nm Amax: 0.10
λ: 405 nm Amax: 0.05
λ: 430-700 nm Amax: 0.01
application(s)
food and beverages
SMILES string
C[N+]([O-])=O
InChI
1S/CH3NO2/c1-2(3)4/h1H3
InChI key
LYGJENNIWJXYER-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- Asymmetric aza-Henry reaction toward trifluoromethyl β-nitroamines and biological investigation of their adamantane-type derivatives.: This study used nitromethane in asymmetric aza-Henry reactions to synthesize trifluoromethyl β-nitroamines, which were further investigated for their biological properties, showcasing the potential of nitromethane in advanced synthetic chemistry (Ren et al., 2024).
- Effect of Temperature on the Liquid Bridging Force while Maintaining Physical Stability in Solid-Liquid Mixed Fuel.: Nitromethane was analyzed in this research to understand its role in the stability of solid-liquid mixed fuels under varying temperatures, providing insights into the optimization of fuel formulations (Zhang et al., 2024).
- Generation of New Synthons for Synthesis Through Activation of Nitromethane.: This research demonstrated the activation of nitromethane to generate new synthons for synthetic applications, highlighting its versatility and importance in creating novel chemical entities (Wang et al., 2024).
- Towards Chemoenzymatic Syntheses of Both Enantiomers of Phosphoemeriamine.: The study explored the use of nitromethane in chemoenzymatic syntheses, enabling the production of both enantiomers of phosphoemeriamine, an important compound in chemical biology (Kiełbasiński et al., 2024).
- Rationally introducing non-canonical amino acids to enhance catalytic activity of LmrR for Henry reaction.: Nitromethane was employed in this study to investigate the enhancement of catalytic activity in the Henry reaction through the introduction of non-canonical amino acids, demonstrating its significance in enzyme catalysis research (Wang et al., 2024).
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Carc. 2 - Flam. Liq. 3 - Repr. 2
Storage Class Code
4.1A - Other explosive hazardous materials
WGK
WGK 2
Flash Point(F)
95.0 °F - closed cup
Flash Point(C)
35 °C - closed cup
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Protocols
GC Analysis of Class 2 Residual Solvents on OVI-G43
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service