W359602
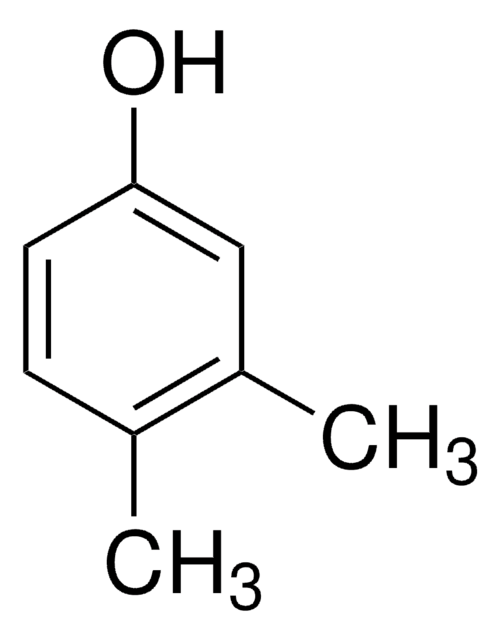
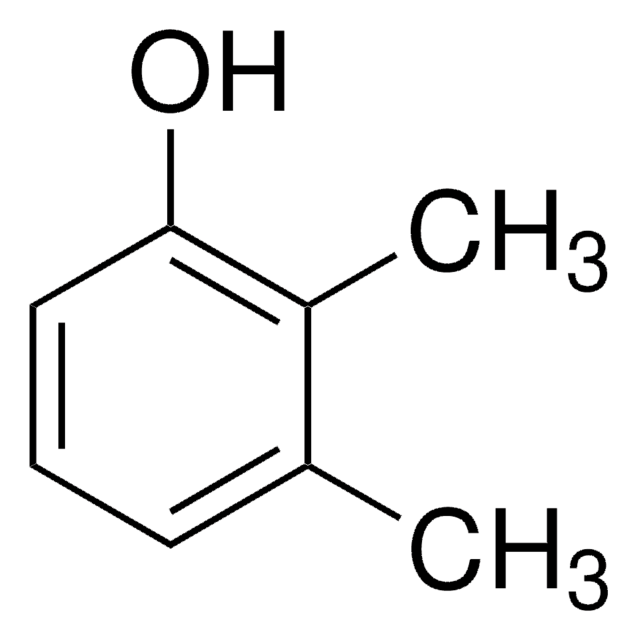
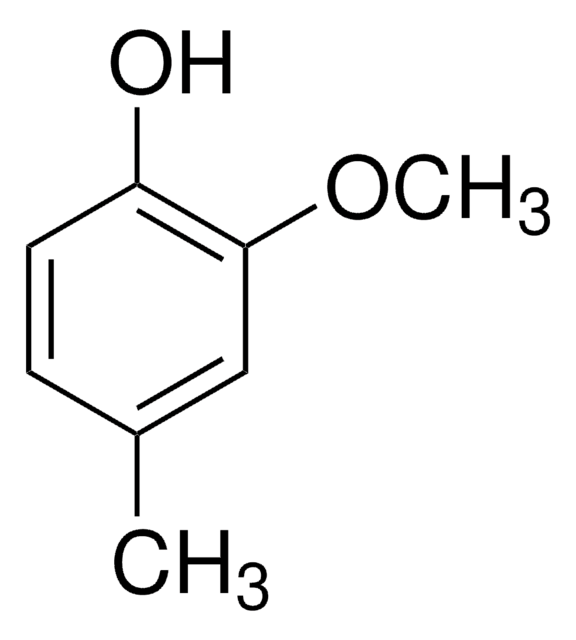
3,4-Xylenol
≥98%, FG
Synonym(s):
3,4-Dimethylphenol, 4-Hydroxy-o-xylene
About This Item
Recommended Products
biological source
synthetic
Quality Level
grade
FG
Fragrance grade
Halal
Agency
follows IFRA guidelines
meets purity specifications of JECFA
reg. compliance
EU Regulation 1223/2009
EU Regulation 1334/2008 & 872/2012
Assay
≥98%
bp
227 °C (lit.)
application(s)
flavors and fragrances
Documentation
see Safety & Documentation for available documents
food allergen
no known allergens
fragrance allergen
no known allergens
Organoleptic
burnt
SMILES string
Cc1ccc(O)cc1C
InChI
1S/C8H10O/c1-6-3-4-8(9)5-7(6)2/h3-5,9H,1-2H3
InChI key
YCOXTKKNXUZSKD-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- Green Aromatic Epoxidation with an Iron Porphyrin Catalyst for One-Pot Functionalization of Renewable Xylene, Quinoline, and Acridine.: This study explores the green epoxidation of aromatic compounds using an iron porphyrin catalyst, demonstrating effective one-pot functionalization of renewable xylene derivatives, including 3,4-xylenol (Corrêa et al., 2023).
- A highly expressed odorant receptor from the yellow fever mosquito, AaegOR11, responds to (+)- and (-)-fenchone and a phenolic repellent.: This research identifies an odorant receptor in mosquitoes that responds to various compounds, including 3,4-dimethylphenol, which could have implications for developing new repellents (Lu et al., 2022).
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 3 Oral - Aquatic Chronic 2 - Eye Dam. 1 - Skin Corr. 1B
Storage Class Code
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 3
Flash Point(F)
230.0 °F - closed cup
Flash Point(C)
110 °C - closed cup
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service