W277002
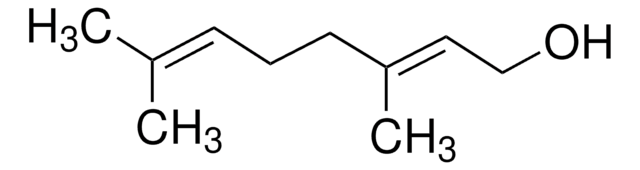
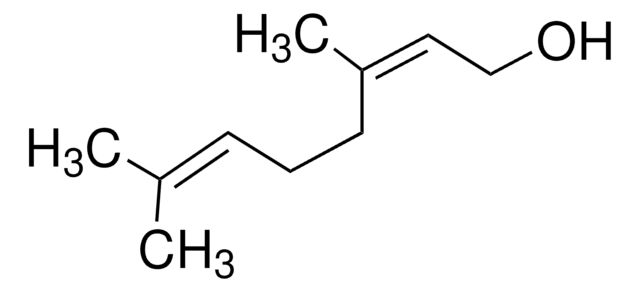
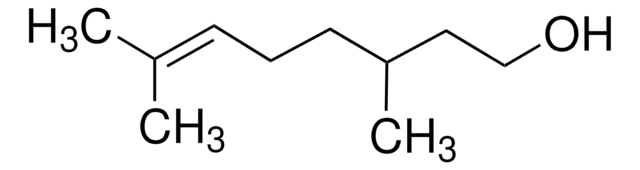
cis-3,7-Dimethyl-2,6-octadien-1-ol
≥97%, FCC, FG
Synonym(s):
Nerol
About This Item
Recommended Products
biological source
synthetic
Quality Level
grade
FG
Fragrance grade
Halal
Kosher
Agency
follows IFRA guidelines
reg. compliance
EU Regulation 1223/2009
EU Regulation 1334/2008 & 178/2002
FCC
FDA 21 CFR 172.515
Assay
≥97%
refractive index
n20/D 1.474 (lit.)
bp
103-105 °C/9 mmHg (lit.)
density
0.876 g/mL at 25 °C (lit.)
application(s)
flavors and fragrances
Documentation
see Safety & Documentation for available documents
food allergen
no known allergens
fragrance allergen
nerol
Organoleptic
fresh; citrus; floral; sweet
SMILES string
C\C(C)=C\CC\C(C)=C/CO
InChI
1S/C10H18O/c1-9(2)5-4-6-10(3)7-8-11/h5,7,11H,4,6,8H2,1-3H3/b10-7-
InChI key
GLZPCOQZEFWAFX-YFHOEESVSA-N
Looking for similar products? Visit Product Comparison Guide
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - Skin Sens. 1B
Storage Class Code
10 - Combustible liquids
WGK
WGK 2
Flash Point(F)
226.0 °F - closed cup
Flash Point(C)
107.78 °C - closed cup
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
-3,7-Dimethyl-2,6-octadien-1-ol; Neral; Geraniol; Geranial; Undecanal; Citronellyl acetate; Neryl acetate; 3,7-Dimethyl-2,6-octadienyl acetate; 1-Tetradecene; Tetradecane; α-Bisabolol
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service