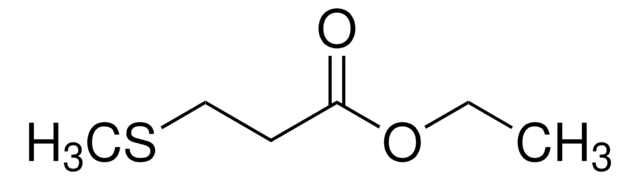
W249300
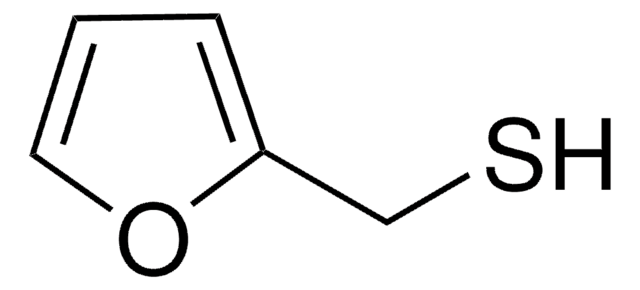
Furfuryl mercaptan
≥97%, FG
Synonym(s):
2-Furanmethanethiol, 2-Furfurylthiol, 2-Furylmethanethiol, Furfuryl mercaptan
About This Item
Recommended Products
biological source
synthetic
Quality Level
grade
FG
Fragrance grade
Halal
Kosher
Agency
follows IFRA guidelines
meets purity specifications of JECFA
reg. compliance
EU Regulation 1223/2009
EU Regulation 1334/2008 & 178/2002
FDA 21 CFR 117
Assay
≥97%
refractive index
n20/D 1.531 (lit.)
bp
155 °C (lit.)
density
1.132 g/mL at 25 °C (lit.)
application(s)
flavors and fragrances
Documentation
see Safety & Documentation for available documents
food allergen
no known allergens
fragrance allergen
no known allergens
Organoleptic
burnt; coffee; meaty; smoky; roasted; sulfurous
SMILES string
SCc1ccco1
InChI
1S/C5H6OS/c7-4-5-2-1-3-6-5/h1-3,7H,4H2
InChI key
ZFFTZDQKIXPDAF-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- Elucidating salt-reduction mechanisms of aroma-active compounds from yeast extracts through sensomics approaches and electroencephalography.: This study investigates the mechanisms by which aroma-active compounds, including furfuryl mercaptan, from yeast extracts reduce saltiness perception. Utilizing sensomics approaches and electroencephalography, the research highlights the potential of these compounds to enhance flavor profiles in food products without increasing sodium content (Shan et al., 2024).
- Comparison of differences in sensory, volatile odour-activity and volatile profile of commercial plant-based meats.: This research compares the sensory and volatile profiles of commercial plant-based meats, identifying furfuryl mercaptan as a key odor-active compound. The findings underscore its role in replicating the savory and meaty aroma characteristic of plant-based meat alternatives (Thong et al., 2024).
- Odorants Identified in Chinese Dry-Cured Ham Contribute to Salty Taste Enhancement.: The study explores how odorants, including furfuryl mercaptan, contribute to the perception of saltiness in Chinese dry-cured ham. This compound′s role in enhancing salty taste highlights its potential application in reducing sodium content in processed meats (Chen et al., 2024).
- Exploring the Effectiveness of Natural Food Flavors in Protecting Carbon Steel against CO(2)-Induced Corrosion.: This research explores the use of natural food flavors, such as furfuryl mercaptan, in protecting carbon steel from CO₂-induced corrosion. The findings suggest potential industrial applications for these compounds in corrosion inhibition (Wang et al., 2023).
Other Notes
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Flam. Liq. 3
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
113.0 °F - closed cup
Flash Point(C)
45 °C - closed cup
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service