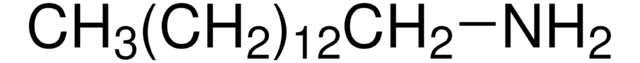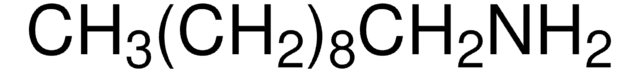All Photos(1)
About This Item
Linear Formula:
CH3(CH2)12NH2
CAS Number:
Molecular Weight:
199.38
Beilstein:
1739990
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
bp
265 °C (lit.)
mp
30-32 °C (lit.)
SMILES string
CCCCCCCCCCCCCN
InChI
1S/C13H29N/c1-2-3-4-5-6-7-8-9-10-11-12-13-14/h2-14H2,1H3
InChI key
ABVVEAHYODGCLZ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Tridecylamine can undergo:
- Dehydrogenation to nitriles catalyzed by ruthenium complex.
- Aerobic oxidation to carbonyl compounds catalyzed by vitamin C and Cu complex.
Tridecylamine is generally used in introducing C13 chain to the substrate. Some of the application are:
- Synthesis of alkylated 1,2,4-triazoles as bridging ligand in the preparation of polymeric 1-dimensional chains of iron(II) species.
- Synthesis of amphiphiles such as N-tridecyl-β-hydroxypropionic acid amide (THPA) and N-(β-hydroxyethyl)tridecanoic acid amide (HETA).
- As a ligand in the preparation of palladium-based catalyst, [PdCl2(TDA)2].
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Eye Dam. 1 - Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
230.0 °F - closed cup
Flash Point(C)
110 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Copper/ascorbic acid dyad as a catalytic system for selective aerobic oxidation of amines
Srogl J and Voltrova S
Organic Letters, 11(4), 843-845 (2009)
Double Dehydrogenation of Primary Amines to Nitriles by a Ruthenium Complex Featuring Pyrazole Functionality
Dutta I, et al.
Journal of the American Chemical Society, 140(28), 8662-8666 (2018)
Spin-transition behaviour in chains of Fe II bridged by 4-substituted 1, 2, 4-triazoles carrying alkyl tails,.
Roubeau O, et al.
New. J. Chem., 25(1), 144-150 (2001)
Catalytic investigation of PdCl2 (TDA)2 immobilized on hydrophobic graphite oxide in the hydrogenation of 1-pentyne and the Heck coupling reaction.
Mastalir A, et al.
Reaction Kinetics, Mechanisms and Catalysis, 113(1), 61-68 (2014)
1-Heptyne semihydrogenation catalized by palladium or rhodium complexes: Influence of: metal atom, ligands and the homo/heterogeneous condition.
Quiroga M E, et al.
Applied Catalysis A: General, 326(2), 121-129 (2007)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service