T33200
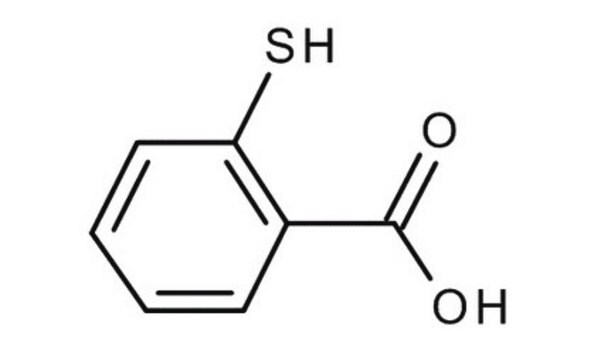
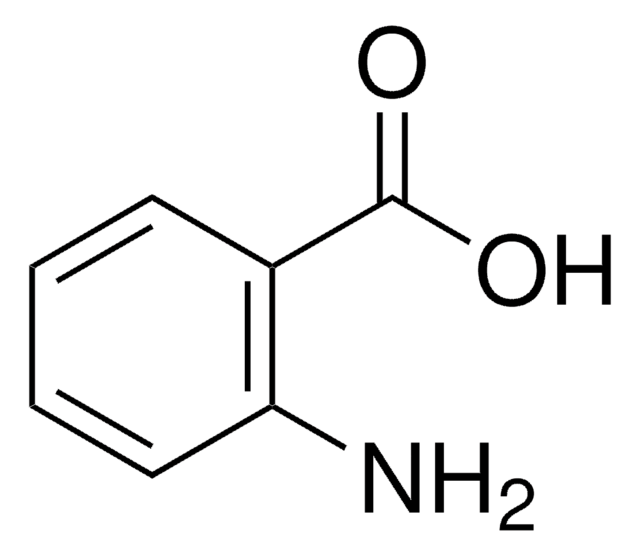
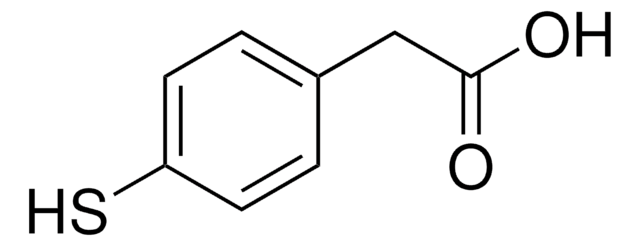
Thiosalicylic acid
97%
Synonym(s):
2-Sulfanylbenzoic acid, 2-Mercaptobenzoic acid
Sign Into View Organizational & Contract Pricing
All Photos(4)
About This Item
Linear Formula:
HSC6H4CO2H
CAS Number:
Molecular Weight:
154.19
Beilstein:
508507
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
mp
162-165 °C (lit.)
SMILES string
OC(=O)c1ccccc1S
InChI
1S/C7H6O2S/c8-7(9)5-3-1-2-4-6(5)10/h1-4,10H,(H,8,9)
InChI key
NBOMNTLFRHMDEZ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Thiosalicylic acid can be used as:
It can also be used to prepare 2-thioxanthone-thioacetic acid bimolecular system, which is used as a photoinitiator for free radical polymerization.
- A nucleophilic trapping agent for the desulfenylation of 3-indolyl sulfides to obtain 3-unsubstituted indoles.
- A starting material to prepare 2′-mercaptoacetophenone, which is used in the synthesis of thioflavanone by reacting with lithium diisopropylamide and benzaldehyde.
- A stabilizing agent in the synthesis of metal nanoparticles.
It can also be used to prepare 2-thioxanthone-thioacetic acid bimolecular system, which is used as a photoinitiator for free radical polymerization.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
The Journal of Organic Chemistry, 59, 6372-6372 (1994)
Synthesis and characterization of thiosalicylic acid stabilized gold nanoparticles
Pattabi RM and Pattabi M
Spectrochimica Acta. Part A, Molecular and Biomolecular Spectroscopy, 74(1), 195-199 (2009)
A new chemical method to desulfenylate indol-3-yl sulfides
Hamel P, et al.
Phosphorus, Sulfur, and Silicon and the Related Elements, 74(1-4), 391-391 (1993)
G Serrano et al.
Journal of the American Academy of Dermatology, 23(3 Pt 1), 479-483 (1990-09-01)
A photocontact dermatitis developed in three patients after the application of gel containing 0.5% piroxicam. Patch tests were positive to thiomersal and thiosalicylic acid. Photopatch tests with piroxicam at several concentrations were positive in the three patients but negative in
One-Component Bimolecular Photoinitiating Systems, 2
Aydin M, et al.
Macromolecular Rapid Communications, 24(12), 718-723 (2003)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service