All Photos(1)
About This Item
Empirical Formula (Hill Notation):
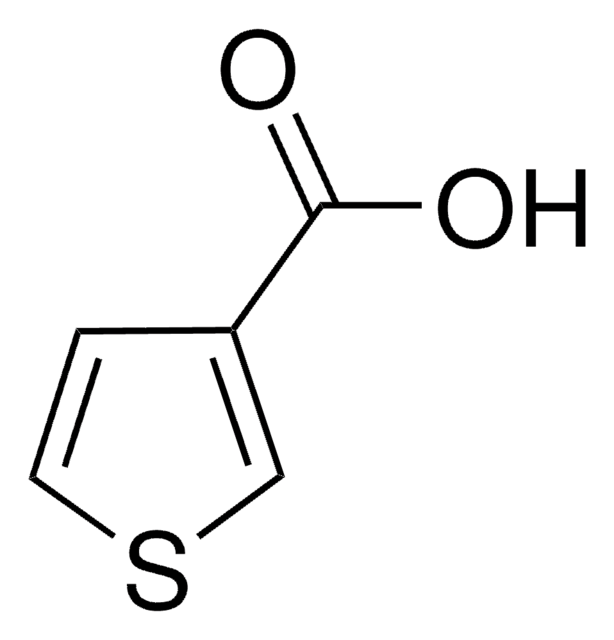
C5H4O2S
CAS Number:
Molecular Weight:
128.15
Beilstein:
110150
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
product line
ReagentPlus®
Assay
99%
bp
260 °C (lit.)
mp
125-127 °C (lit.)
SMILES string
OC(=O)c1cccs1
InChI
1S/C5H4O2S/c6-5(7)4-2-1-3-8-4/h1-3H,(H,6,7)
InChI key
QERYCTSHXKAMIS-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Legal Information
ReagentPlus is a registered trademark of Merck KGaA, Darmstadt, Germany
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Matthew F McCown et al.
Antimicrobial agents and chemotherapy, 53(5), 2129-2132 (2009-03-11)
In vitro, telaprevir selects subtype-specific resistance pathways for hepatitis C virus GT-1a and GT-1b, as described to have occurred in patients. In GT-1a, the HCV-796 resistance mutation C316Y has low replication capacity (7%) that can be compensated for by the
Laval Chan et al.
Bioorganic & medicinal chemistry letters, 14(3), 797-800 (2004-01-27)
Further SAR studies on the thiophene-2-carboxylic acids are reported. These studies led to the identification of a series of tertiary amides that show inhibition of both HCV NS5B polymerase in vitro and HCV subgenomic RNA replication in Huh-7 cells. Structural
Kerim Babaoglu et al.
Nature chemical biology, 2(12), 720-723 (2006-10-31)
Fragment-based screens test multiple low-molecular weight molecules for binding to a target. Fragments often bind with low affinities but typically have better ligand efficiencies (DeltaG(bind)/heavy atom count) than traditional screening hits. This efficiency, combined with accompanying atomic-resolution structures, has made
E Streit et al.
The European respiratory journal, 4(6), 718-722 (1991-06-01)
A randomised double-blind crossover study compared the clinical effectiveness of a 21-day treatment with 600 mg per day of sodium thiophene carboxylate and placebo in 33 patients with stable chronic bronchial disease. During the seven week trial, subjective symptoms and
Salvatore Profeta et al.
Journal of molecular graphics & modelling, 28(6), 540-547 (2010-01-12)
In the course of investigating the propensity of aromatic acids to react with selected nucleophiles, we came across an interesting difference in yields for two structurally similar thiophene carboxylic acids. Given that these yields were consistent across more than 40
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)