901116
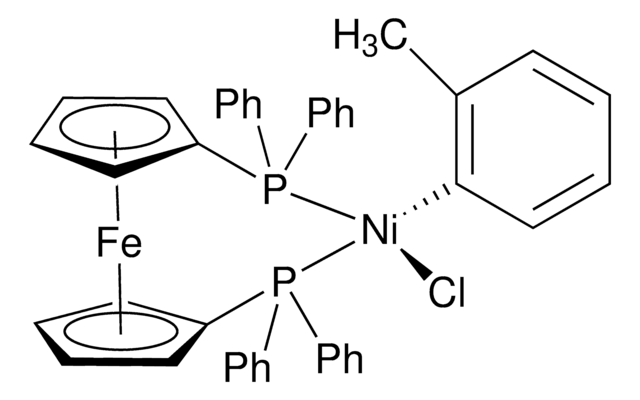
trans-Bis(dicyclohexylphenylphosphine)(2-methylphenyl)nickel(II) chloride
Synonym(s):
trans-(PCy2Ph)2Ni(o-tolyl)Cl
About This Item
Recommended Products
form
powder or solid
reaction suitability
core: nickel
reaction type: Cross Couplings
reagent type: catalyst
mp
174-179 °C
SMILES string
Cl[Ni](P(C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C1([H])[H])(C2=C([H])C([H])=C([H])C([H])=C2[H])C3([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C3([H])[H])(P(C4([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C4([H])[H])(C5=C([H])C([H])=C([H
Other Notes
Legal Information
related product
Storage Class Code
13 - Non Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
The Jamison group has developed a library of bench-stable phosphine-containing nickel(II) precatalysts that are converted into active catalysts in situ.
Related Content
Research in the Jamison group is centered on the development of new reactions and technologies for organic synthesis. Towards these themes, the group has pioneered a number of air-stable nickel precatalysts supported by phosphines and N-heterocyclic carbenes that are readily converted to the active catalyst in situ.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service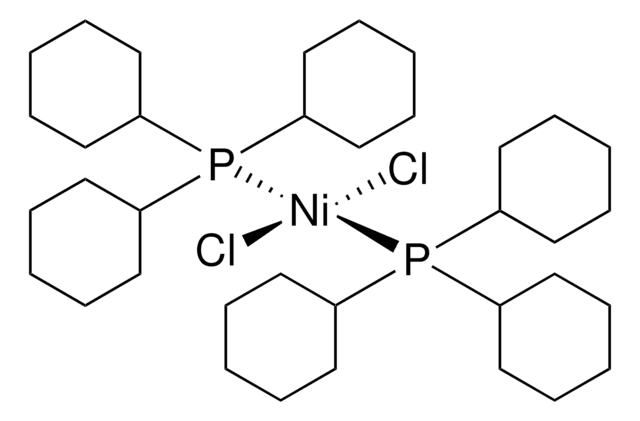
nickel(II) chloride](/deepweb/assets/sigmaaldrich/product/structures/252/197/3c560697-beb3-4c52-85d6-ebc3af13db69/640/3c560697-beb3-4c52-85d6-ebc3af13db69.png)
![[(TMEDA)Ni(o-tolyl)Cl] 95%](/deepweb/assets/sigmaaldrich/product/structures/236/439/768c916e-994f-47e3-a980-3ca0471317d7/640/768c916e-994f-47e3-a980-3ca0471317d7.png)
![[1,2-Bis(diphenylphosphino)ethane]dichloronickel(II)](/deepweb/assets/sigmaaldrich/product/structures/707/956/483e7a6e-5fb5-4e39-abd1-ecf33ccab3cf/640/483e7a6e-5fb5-4e39-abd1-ecf33ccab3cf.png)
![[1,1′-Bis(diphenylphosphino)ferrocene]dichloronickel(II) 97%](/deepweb/assets/sigmaaldrich/product/structures/274/566/a60d6584-163a-4c41-a738-60f8e4d524fa/640/a60d6584-163a-4c41-a738-60f8e4d524fa.png)