900755
SLAP TA
95%
Synonym(s):
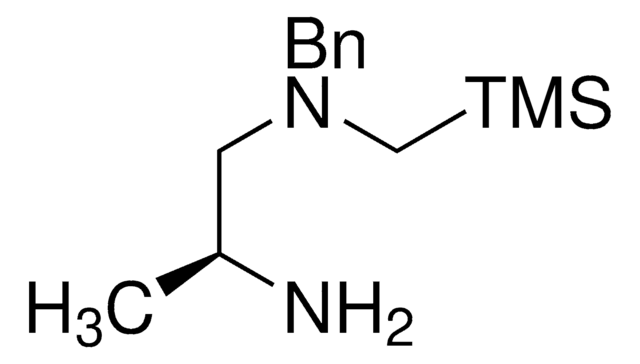
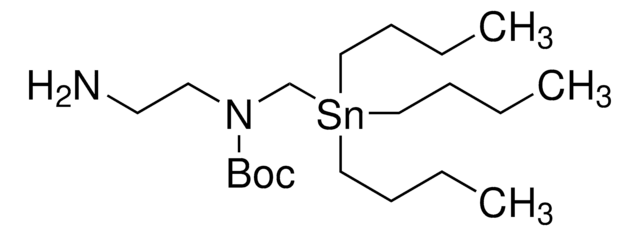
3-(((Trimethylsilyl)methyl)thio)propan-1-amine
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C7H19NSSi
CAS Number:
Molecular Weight:
177.38
UNSPSC Code:
12352103
NACRES:
NA.22
Recommended Products
Related Categories
Application
When used with aliphatic or aromatic aldehydes, this silicon amine protocol SLAP reagent enables the photomediated synthesis of thiomorpholines and thiazepanes. For photocatalytic cross-coupling with this SLAP Reagent, Ir[(ppy)2dtbbpy]PF6 (747769) is used with a recommended starting combination of Lewis acids Bi(OTf)3 (633305) and Cu(OTf)2 (283673) prior to subsequent substrate-specific optimization. This product was introduced in collaboration with the Bode Research Group and provides a tin-free alternative to SnAP (tin amine protocol) reagents and is well-suited for scale-up reactions.
Other Notes
- Technology Spotlight: SLAP Reagents for Piperazine Synthesis
- Silicon Amine Reagents for the Photocatalytic Synthesis of Piperazines from Aldehydes and Ketones
- Lewis Acid Induced Toggle from Ir(II) to Ir(IV) Pathways in Photocatalytic Reactions: Synthesis of Thiomorpholines and Thiazepanes from Aldehydes and SLAP Reagents.
- Continuous Flow Synthesis of Morpholines and Oxazepanes with Silicon Amine Protocol (SLAP) Reagents and Lewis Acid Facilitated Photoredox Catalysis
related product
Product No.
Description
Pricing
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Flam. Liq. 3
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
75.0 °F
Flash Point(C)
23.89 °C
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Moritz K Jackl et al.
Organic letters, 19(17), 4696-4699 (2017-08-17)
Photocatalytic coupling of aldehydes and silicon amine protocol (SLAP) reagents enables the simple, scalable synthesis of substituted morpholines, oxazepanes, thiomorpholines, and thiazepanes under continuous flow conditions. Key to the success of this process is the combination of an inexpensive organic
Sheng-Ying Hsieh et al.
Organic letters, 18(9), 2098-2101 (2016-04-22)
Silicon amine protocol (SLAP) reagents for photocatalytic cross-coupling with aldehydes and ketones to form N-unprotected piperazines have been developed. This blue light promoted process tolerates a wide range of heteroaromatic, aromatic, and aliphatic aldehydes and structurally and stereochemically complex SLAP
Sheng-Ying Hsieh et al.
ACS central science, 3(1), 66-72 (2017-02-06)
Redox neutral photocatalytic transformations often require careful pairing of the substrates and photoredox catalysts in order to achieve a catalytic cycle. This can limit the range of viable transformations, as we recently observed in attempting to extend the scope of
Protocols
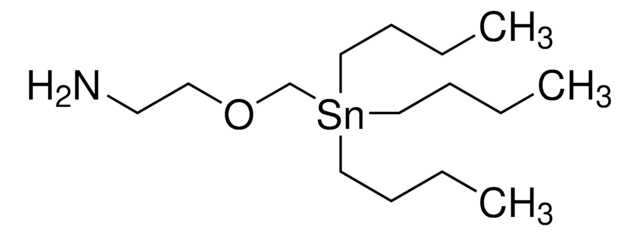
The Bode group has developed SnAP (stannyl amine protocol) reagents that cross-couple with aldehydes and ketones to provide one-step access to a wide variety of saturated N-heterocycles.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service