796832
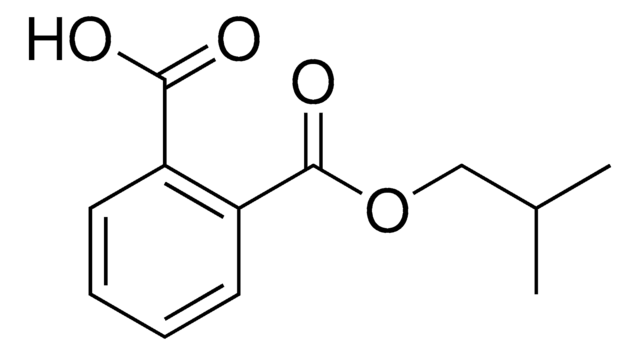
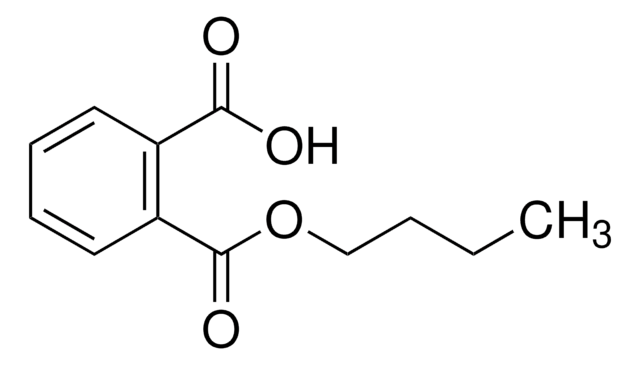
Phthalic acid mono-2-ethylhexyl ester
97%
Synonym(s):
mono-2-Ethylhexyl phthalate
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C16H22O4
CAS Number:
Molecular Weight:
278.34
Beilstein:
3206630
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
description
Flash Point: >110°C
Quality Level
Assay
97%
form
liquid (Colorless)
refractive index
n/D 1.5051
density
1.0864 g/mL at 25 °C
SMILES string
O=C(O)C1=CC=CC=C1C(OCC(CC)CCCC)=O
InChI
1S/C16H22O4/c1-3-5-8-12(4-2)11-20-16(19)14-10-7-6-9-13(14)15(17)18/h6-7,9-10,12H,3-5,8,11H2,1-2H3,(H,17,18)
InChI key
DJDSLBVSSOQSLW-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Phthalic acid mono-2-ethylhexyl ester can be used as a precursor:
- To synthesize dioctyl phthalate, a widely used plasticizer, via esterification reaction with 2-ethylhexanol.
- To prepare a fluorescent probe, 2-[[(3′,6′-dihydroxy-3-oxospiro[isobenzofuran-1(3H),9′-[9H]xanthen]-5-yl)amino]carbonyl] 2-ethylhexyl benzoic acid ester, to detect toxic metabolite mono-2-ethylhexyl phthalate (MEHP).
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Reactive Extraction Strategy for Synthesizing Dioctyl Phthalate Intensified by Bifunctional Deep Eutectic Solvent [Im: 2PTSA]
Qin Hao, et al.
Chemical Engineering and Processing, 157, 108060-108060 (2020)
Da-Hye Kim et al.
Molecules (Basel, Switzerland), 24(8) (2019-04-24)
Endocrine active compounds with structural similarities to natural hormones such as 17β-estradiol (E2) and androgen are suspected to affect the human endocrine system by inducing hormone-dependent effects. This study aimed to detect the (anti-)estrogenic and (anti-)androgenic activities of mono-(2-ethylhexyl) phthalate
Chunjiao Lu et al.
Ecotoxicology and environmental safety, 208, 111525-111525 (2020-10-30)
The base excision repair (BER) pathway is an important defense response to oxidative DNA damage. It is known that exposures to phthalate esters (PAEs), including Dibutyl phthalate (DBP), Mono-(2-ethylhexyl) phthalate (MEHP), and Di-(2-ethylhexyl) phthalate (DEHP), cause reactive oxygen species-induced DNA
Jijun Shang et al.
Toxicological sciences : an official journal of the Society of Toxicology, 168(1), 78-94 (2018-11-07)
Bisphenols and phthalates leach from medical devices, and this exposure is likely to increase in postcardiac surgery patients. Previous studies suggest that such chemical exposure may impact recovery and wound healing, yet the direct effects of bisphenols and phthalates are
Xiaoju Ma et al.
Toxicology research, 8(4), 522-530 (2019-08-02)
Recent reports have concentrated on some androgens/antiandrogens and confirmed that certain chemicals have demonstrated androgenic/antiandrogenic activities in vitro. However, it is still unknown whether more chemicals in the human environment possess endocrine toxicity. 58A novel AR-mediated reporter gene assay based
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service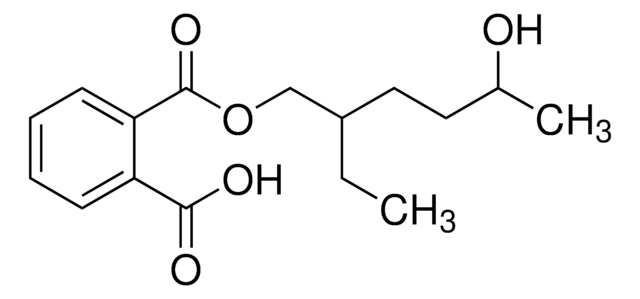
![mono-[(2RS)-2-Ethyl-5-oxohexyl] phthalate analytical standard](/deepweb/assets/sigmaaldrich/product/structures/380/975/47f8fcbc-594c-4f9c-84df-43685552cd06/640/47f8fcbc-594c-4f9c-84df-43685552cd06.png)