All Photos(1)
About This Item
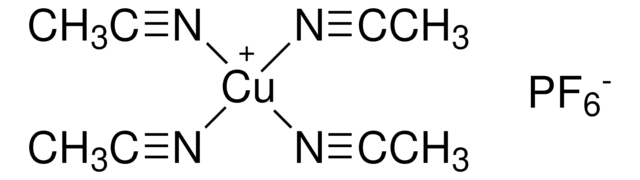
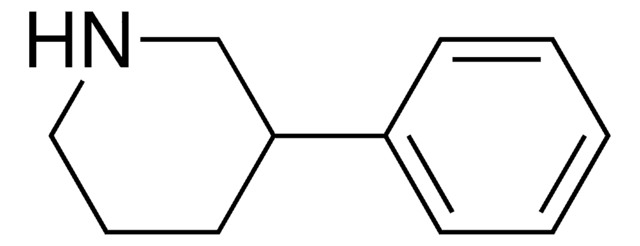
Empirical Formula (Hill Notation):
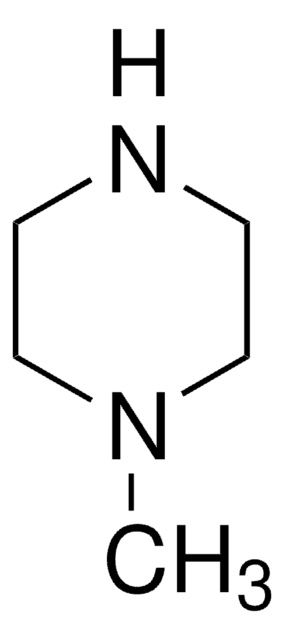
C11H15N
CAS Number:
Molecular Weight:
161.24
Beilstein:
124508
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
mp
61-65 °C (lit.)
SMILES string
C1CC(CCN1)c2ccccc2
InChI
1S/C11H15N/c1-2-4-10(5-3-1)11-6-8-12-9-7-11/h1-5,11-12H,6-9H2
InChI key
UTBULQCHEUWJNV-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
G H Loew et al.
Molecular pharmacology, 34(3), 363-376 (1988-09-01)
The 4-(m-OH-phenyl)piperidines are a flexible fragment of the morphine/benzomorphan fused-ring opioids. Analogs in this family were synthesized with varying 4-alkyl substituents increasing in bulk from H through methyl, n-propyl, to t-butyl, each with the three N-substituents methyl, allyl, and phenethyl.
B L Blaylock et al.
Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology, 36(5), 1104-1113 (2011-02-04)
Although dopamine D(3) receptors have been associated with cocaine abuse, little is known about the consequences of chronic cocaine on functional activity of D(3) receptor-preferring compounds. This study examined the behavioral effects of D(3) receptor-selective 4-phenylpiperazines with differing in vitro
P Singh et al.
Journal of enzyme inhibition, 16(4), 331-338 (2002-03-28)
Two series of compounds were recently reported as novel alpha1a-selective adrenoceptor antagonists. In the first series, a dihydropyrimidone moiety is attached to a 4-phenyl piperidine containing side chain, while in the second, it is linked to a 4-substituted phenyl piperazine
Xing-hai Wang et al.
European journal of medicinal chemistry, 41(2), 226-232 (2006-01-13)
A nonlinear QSAR study was conducted on a series of 4-phenylpiperidine derivatives (4PPs) acting as mu opioid agonists by three-layer back-propagation neural network (NN) method. At first a variety of molecular descriptors were calculated and then selected with two-stage least
Karel Vervisch et al.
Organic & biomolecular chemistry, 10(16), 3308-3314 (2012-03-14)
Non-activated 2-(4-chloro-2-cyano-2-phenylbutyl)aziridines were used as building blocks for the stereoselective synthesis of novel cis-2-cyanomethyl-4-phenylpiperidines via a microwave-assisted aziridine to piperidine ring expansion followed by a radical-induced nitrile translocation through initial formation and subsequent cleavage of intermediate bicyclic iminyl radicals. Furthermore
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service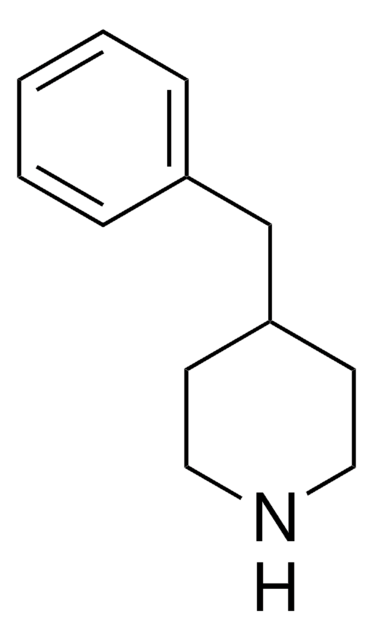

![Benzo[b]thien-2-ylboronic acid ≥95%](/deepweb/assets/sigmaaldrich/product/structures/251/077/d0ead874-b533-4dcb-890d-8816a0018ccd/640/d0ead874-b533-4dcb-890d-8816a0018ccd.png)

![9-Azabicyclo[3.3.1]nonane N-oxyl 95%](/deepweb/assets/sigmaaldrich/product/structures/287/155/e2f4a2e1-1d4e-4bed-9187-9e16d23cbbbf/640/e2f4a2e1-1d4e-4bed-9187-9e16d23cbbbf.png)