460923
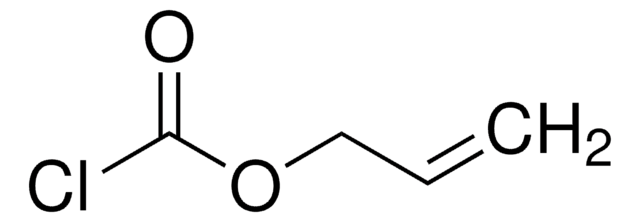
Propargyl chloroformate
96%
Synonym(s):
(Prop-2-ynyloxy)carbonyl chloride, 2-Propyn-1-yl carbonochloridate, 2-Propynyl chloroformate, Prop-2-yn-1-yl carbonochloridate, Propargyloxycarbonyl chloride
About This Item
Recommended Products
vapor pressure
0.65 psi ( 20 °C)
Quality Level
Assay
96%
reaction suitability
reaction type: click chemistry
refractive index
n20/D 1.435 (lit.)
bp
118-122 °C (lit.)
density
1.215 g/mL at 25 °C (lit.)
storage temp.
2-8°C
SMILES string
ClC(=O)OCC#C
InChI
1S/C4H3ClO2/c1-2-3-7-4(5)6/h1H,3H2
InChI key
RAMTXCRMKBFPRG-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- binaphthol-diacetylene derivatives
- propargyl pentafluorophenyl carbonate
- N-Poc protected amino acids (Poc = propargyloxycarbonyl)
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Flam. Liq. 3 - Skin Corr. 1B
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
86.0 °F - closed cup
Flash Point(C)
30 °C - closed cup
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service