All Photos(1)
About This Item
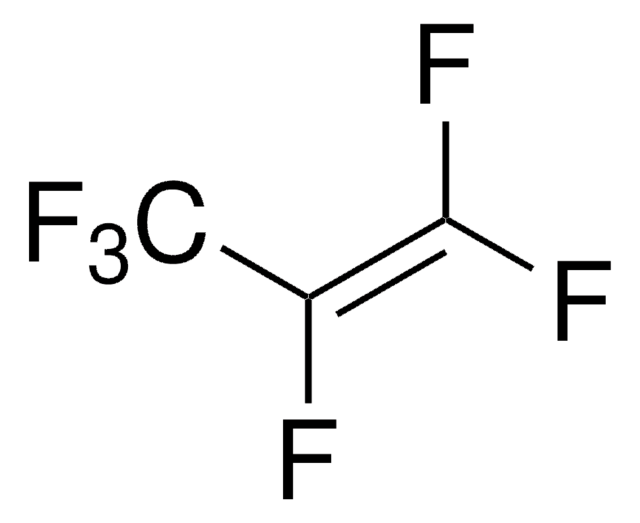
Linear Formula:
[CF3C(F)=CF2]3
CAS Number:
Molecular Weight:
450.07
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
form
liquid
Quality Level
bp
110-115 °C (lit.)
density
1.83 g/mL at 25 °C (lit.)
SMILES string
FC(F)=C(F)C(F)(F)F.FC(F)=C(F)C(F)(F)F.FC(F)=C(F)C(F)(F)F
InChI
1S/3C3F6/c3*4-1(2(5)6)3(7,8)9
InChI key
VJRSWIKVCUMTFK-UHFFFAOYSA-N
General description
Spectra of three isomeric trimers of hexafluoropropene have been investigated. It reacts with primary amines, via indirect substitution of fluorine atoms, to form the corresponding enamines and enimines.
Application
Hexafluoropropene, trimer may be used in the preparation of photochromic spiroindolinonaphthoxazine derivative. It may be used in the preparation of 2-trifluoromethyl-3-heptadifluoroisopropyl-2-perfluoropentene-4-(N′,N′-dimethylamino propyl) imino(I).
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Irrit. 2 - Flam. Liq. 3 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
75.2 °F - closed cup
Flash Point(C)
24 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
C C Geilen et al.
FEBS letters, 309(3), 381-384 (1992-09-14)
In this study, we report that the potent and selective inhibitor of cyclic AMP-dependent protein kinase, N-[2-bromocinnamyl(amino)ethyl]-5-isoquinolinesulfonamide (H-89) interferes with the incorporation of choline into phosphatidylcholine in HeLa cells. Treatment of cells with 10 microM H-89 for 1 h decreases
W L Combest et al.
Journal of neurochemistry, 51(5), 1581-1591 (1988-11-01)
The effects of the naturally occurring polyamines spermine and spermidine on phosphorylation promoted by cyclic AMP (cAMP)-dependent protein kinase (PK) (cAMP-PK; EC 2.7.1.37) were studied using the brain of the tobacco hornworm, Manduca sexta. Four particulate-associated peptides (280, 34, 21
H Kawasaki et al.
Science (New York, N.Y.), 282(5397), 2275-2279 (1998-12-18)
cAMP (3',5' cyclic adenosine monophosphate) is a second messenger that in eukaryotic cells induces physiological responses ranging from growth, differentiation, and gene expression to secretion and neurotransmission. Most of these effects have been attributed to the binding of cAMP to
Synthesis of Fluoro-tertiary and Quaternary Ammonium Cationic Surfactants [J].
Jingfeng LZ, et al.
Dyestuffs and Coloration / Ran Liao Yu Ran Shai, 1, 009-009 (2006)
Jost Leemhuis et al.
The Journal of pharmacology and experimental therapeutics, 300(3), 1000-1007 (2002-02-28)
The small GTPase RhoA can retract cell extensions by acting on two Rho kinases (ROCKs). Activated protein kinase A (PKA) inhibits RhoA and induces extensions. The isoquinoline H89 inhibits PKA and thus should prevent the inactivation of RhoA. In kinase
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service