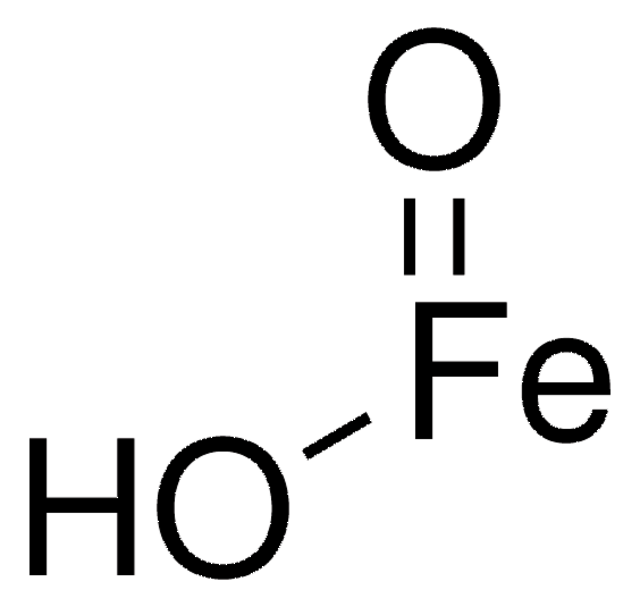
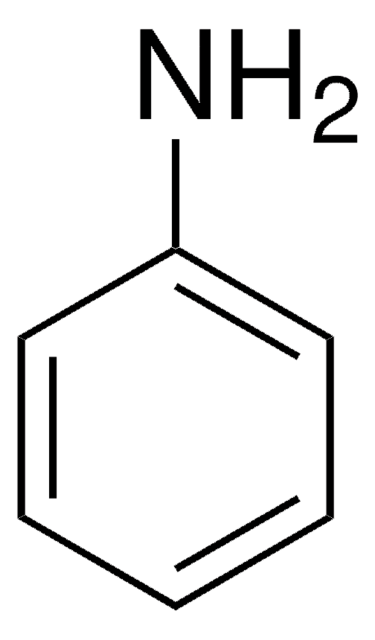
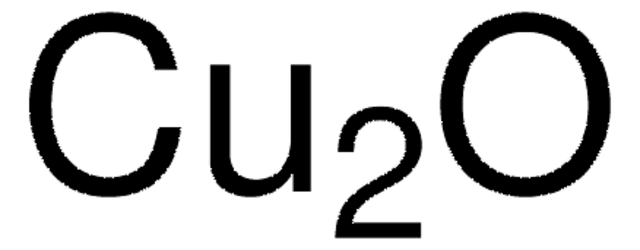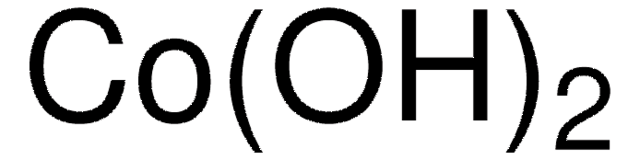289787
Copper(II) hydroxide
technical grade
Synonym(s):
Cupric hydroxide
About This Item
Recommended Products
grade
technical grade
Quality Level
form
powder
contains
stabilizer
concentration
≥57.0% Cu (EDTA titration)
solubility
H2O: insoluble(lit.)
aqueous acid: slightly soluble(lit.)
application(s)
battery manufacturing
SMILES string
O[Cu]O
InChI
1S/Cu.2H2O/h;2*1H2/q+2;;/p-2
InChI key
JJLJMEJHUUYSSY-UHFFFAOYSA-L
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 2 Inhalation - Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Eye Dam. 1
Storage Class Code
6.1B - Non-combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Thermoelectric Performance of Perovskite-type Oxide Materials
The prevailing strategies for heat and electric-power production that rely on fossil and fission fuels are having a negative impact on the environment and on our living conditions.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service