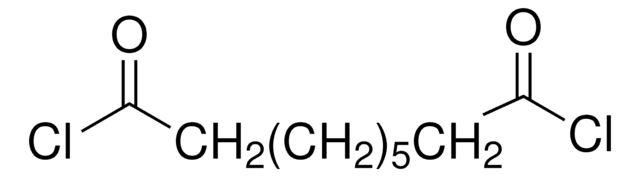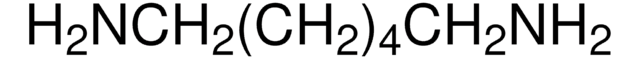All Photos(1)
About This Item
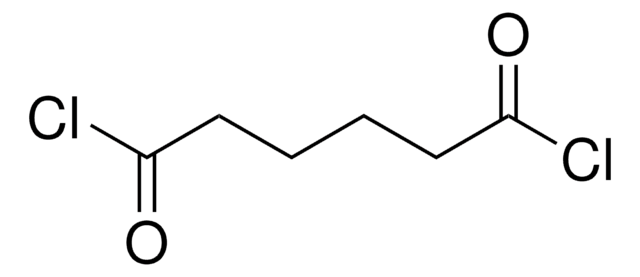
Linear Formula:
ClCO(CH2)5COCl
CAS Number:
Molecular Weight:
197.06
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
refractive index
n20/D 1.469 (lit.)
bp
113 °C/5 mmHg (lit.)
density
1.205 g/mL at 25 °C (lit.)
SMILES string
ClC(=O)CCCCCC(Cl)=O
InChI
1S/C7H10Cl2O2/c8-6(10)4-2-1-3-5-7(9)11/h1-5H2
InChI key
LVIMBOHJGMDKEJ-UHFFFAOYSA-N
Application
Pimeloyl chloride has been used in preparation of:
- bis-acylureas, by the coupling reaction with ureas
- triacetyl-15-pimelate-nivalenol, required for production of antibody against nivalenol tetraacetate
- macrocyclic tetralactones, via condensation with stannoxane
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1 - Skin Corr. 1B - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
235.4 °F - closed cup
Flash Point(C)
113 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Tin derivatives for synthesis: the preparation of macrocyclic lactones.
Shanzer A and Mayer-Shochet N.
Journal of the Chemical Society. Chemical Communications, 4, 176-177 (1980)
Jong-Uk Kim et al.
Journal of colloid and interface science, 359(2), 428-435 (2011-05-04)
We present the synthesis and morphology study of thirteen bis-acylurea molecules with various functional end groups. The bis-acylureas have two acylurea groups, -NH-CO-NH-CO-, divided by a pentamethylene spacer, -(CH(2))(5)-, and two symmetric functional end groups, such as, aliphatic, benzyl, mono-
C R Wang et al.
Applied and environmental microbiology, 57(4), 1026-1030 (1991-04-01)
Antibody against nivalenol tetraacetate (tetra-Ac-NIV) was prepared by immunization of rabbits with triacetyl-15-pimelate-NIV conjugated to bovine serum albumin. By using tritiated tetra-Ac-NIV as the test ligand, antibody titers were demonstrated as early as 4 weeks after immunization. Useful antibody for
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service