All Photos(1)
About This Item
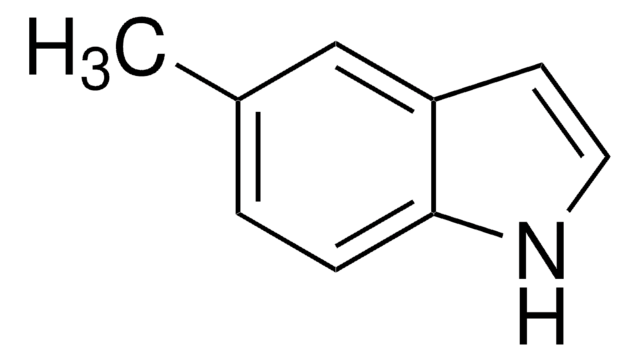
Empirical Formula (Hill Notation):
C9H9N
CAS Number:
Molecular Weight:
131.17
Beilstein:
109708
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
form
solid
refractive index
n20/D 1.607 (lit.)
bp
112 °C/5 mmHg (lit.)
density
1.059 g/mL at 25 °C (lit.)
SMILES string
Cc1ccc2cc[nH]c2c1
InChI
1S/C9H9N/c1-7-2-3-8-4-5-10-9(8)6-7/h2-6,10H,1H3
InChI key
ONYNOPPOVKYGRS-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
6-Methylindole was used in the synthesis of benz[c,d]indol-3(1H)-one derivatives.
- Reactant for preparation of tryptophan dioxygenase inhibitors pyridyl-ethenyl-indoles as potential anticancer immunomodulators
- Reactant for preparation of indole linked triazole derivatives as antifungal agents
- Reactant for preparation of N-β-D-xylosyl-indole derivatives as SGLT2 inhibitors for management of hyperglycemia in diabetes
- Reactant for preparation of aminoguanidine derivatives of arylsulfonylacylindoles as antifungal agents
- Reactant for preparation of indolylindazoles and indolylpyrazolopyridines as interleukin-2 inducible T cell kinase inhibitors
- Reactant for preparation of arylsulfonylacetylindoles as anti-HIV-1 agents
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
230.0 °F - closed cup
Flash Point(C)
110 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Taichi Komoda et al.
Bioscience, biotechnology, and biochemistry, 67(3), 659-662 (2003-05-02)
The new inhibitors of 3alpha-hydroxysteroid dehydrogenase, 0231A 1 and 0231B 2, have a unique benz[c,d]indol-3(1H)-one structure in their molecules. In our advanced studies on indole chemistry, we have developed an efficient synthetic method for benz[c,d]indol-3(1H)-one derivatives. We report here its
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service