All Photos(1)
About This Item
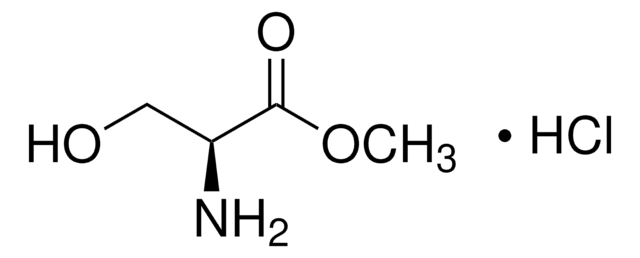
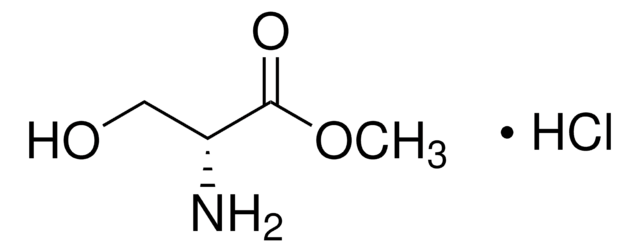
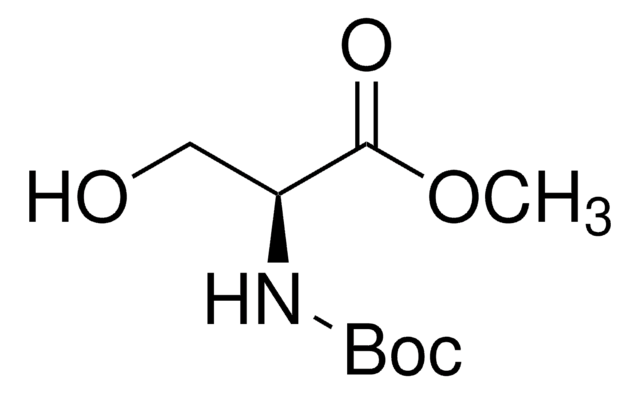
Linear Formula:
HOCH2CH(NH2)CO2CH3·HCl
CAS Number:
Molecular Weight:
155.58
EC Number:
MDL number:
UNSPSC Code:
12352209
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
form
powder
reaction suitability
reaction type: solution phase peptide synthesis
mp
134-136 °C (lit.)
application(s)
peptide synthesis
SMILES string
Cl[H].COC(=O)C(N)CO
InChI
1S/C4H9NO3.ClH/c1-8-4(7)3(5)2-6;/h3,6H,2,5H2,1H3;1H
InChI key
NDBQJIBNNUJNHA-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
J Sélambarom et al.
Carbohydrate research, 330(1), 43-51 (2001-02-24)
The reaction of L-serine methyl ester hydrochloride (1) with paraformaldehyde (2) in dichloromethane in the presence of triethylamine afforded a novel compound: [lS,2S,6S,7S]-1,6-diaza-4,9-dioxa-2,7-dimethoxycarbonylbicyclo[4.4.1]undecane (4) as a 2:3 adduct of 1 with 2. 1H and 13C NMR spectroscopy were unable to
Cédric Couturier et al.
Organic letters, 8(10), 2183-2186 (2006-05-05)
[reaction: see text] Reaction of N,N-dibenzyl-O-methylsulfonyl serine methyl ester with a variety of heteronucleophiles (sodium azide, sodium phthalimide, amines, thiols) and carbanions (sodium malonate) gave, via an aziridinium intermediate, the corresponding beta-amino or alpha,beta-diamino ester in good to excellent yield.
Acylated serine derivatives: a unique class of arthropod pheromones of the Australian redback spider, Latrodectus hasselti.
Elena Jerhot et al.
Angewandte Chemie (International ed. in English), 49(11), 2037-2040 (2010-02-11)
Yu Harayama et al.
Chemical communications (Cambridge, England), (13)(13), 1764-1766 (2005-03-26)
The use of hypervalent iodine(III) reagents allowed us to develop the novel and efficient direct synthesis of N,O-acetal compounds via the oxidative fragmentation reaction of alpha-amino acids or alpha-amino alcohols; furthermore, we succeeded in developing an improved synthesis of the
Carlos Aydillo et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 13(17), 4840-4848 (2007-03-17)
A new chiral serine equivalent and its enantiomer have been synthesized from (S)- and (R)-N-Boc-serine methyl esters (Boc: tert-butyloxycarbonyl). The use of these compounds as chiral building blocks has been demonstrated in the synthesis of alpha-alkyl alpha-amino acids by diastereoselective
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service