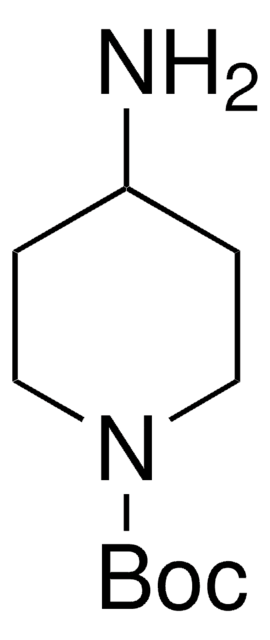
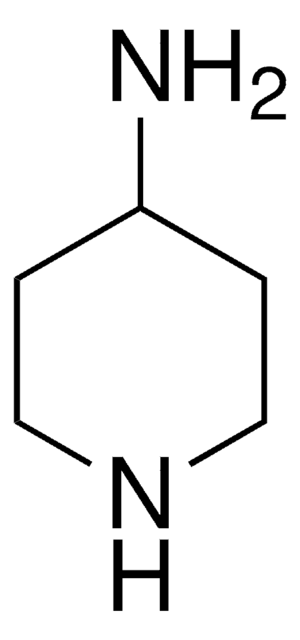
198064
Ethyl 4-amino-1-piperidinecarboxylate
96%
Synonym(s):
1-Carbethoxy-4-aminopiperidine
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
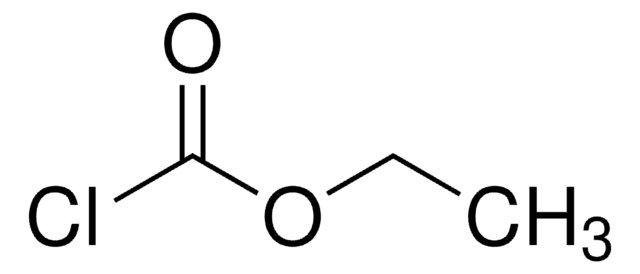
C8H16N2O2
CAS Number:
Molecular Weight:
172.22
Beilstein:
388597
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
96%
form
liquid
refractive index
n20/D 1.483 (lit.)
density
1.004 g/mL at 25 °C (lit.)
SMILES string
CCOC(=O)N1CCC(N)CC1
InChI
1S/C8H16N2O2/c1-2-12-8(11)10-5-3-7(9)4-6-10/h7H,2-6,9H2,1H3
InChI key
GQQQULCEHJQUJT-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
Ethyl 4-amino-1-piperidinecarboxylate (1-Carbethoxy-4-aminopiperidine) was used in the synthesis of quinolin-2(1H )-one derivatives.
Reactant for synthesis of:
Milrinone analogs to analyze effects on intracellular calcium increase in cardiac cells
Tetrahydronaphthalene derivatives
Quorum sensing modulators
Selective anti-Helicobacter pylori activity molecules
Orally active M1 mAChR agonists
Aza derivatives of phytoalexin for antibacterial activity
Milrinone analogs to analyze effects on intracellular calcium increase in cardiac cells
Tetrahydronaphthalene derivatives
Quorum sensing modulators
Selective anti-Helicobacter pylori activity molecules
Orally active M1 mAChR agonists
Aza derivatives of phytoalexin for antibacterial activity
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
172.4 °F - closed cup
Flash Point(C)
78 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Christopher Blackburn et al.
Bioorganic & medicinal chemistry letters, 16(10), 2621-2627 (2006-03-10)
Several potent, functionally active MCHr1 antagonists derived from quinolin-2(1H)-ones and quinazoline-2(1H)-ones have been synthesized and evaluated. Pyridylmethyl substitution at the quinolone 1-position results in derivatives with low-nM binding potency and good selectivity with respect to hERG binding.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service