All Photos(2)
About This Item
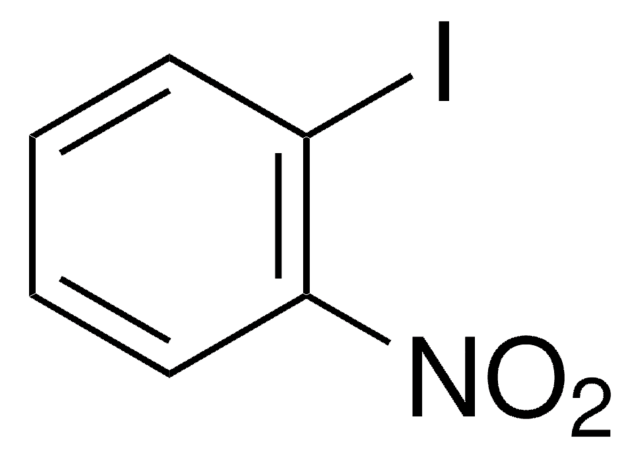
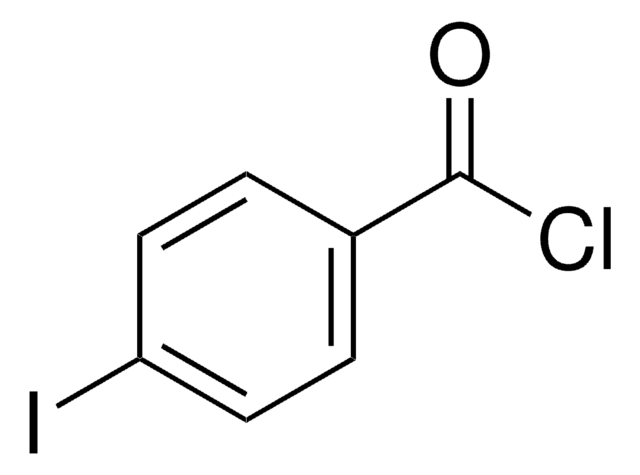
Linear Formula:
IC6H4CH2Cl
CAS Number:
Molecular Weight:
252.48
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
form
powder
refractive index
n20/D 1.635 (lit.)
bp
147-149 °C/32 mmHg (lit.)
mp
29-31 °C (lit.)
SMILES string
ClCc1ccccc1I
InChI
1S/C7H6ClI/c8-5-6-3-1-2-4-7(6)9/h1-4H,5H2
InChI key
FTMNWZHKQGKKAU-UHFFFAOYSA-N
Related Categories
Application
2-Iodobenzyl chloride was used in the synthesis of:
- succinimido[3,4-b]indane
- 1,2,3,4,5,6-hexahydro-1,5-methano-3-benzazocine-2,4-dione
- 1-(2-iodobenzyi)-1,1-diethylamine
- 1-(2-iodobenzyl)pyrrolidine
- 1-(2-iodobenzyl)piperidine
- 1-(2-iodobenzyl)hexahydroazepine
- 4-(2-iodobenzyl)morpholine
- 4-(2-iodobenzyl)thiomorpholine
- 1-benzoyl-4-(2-iodobenzyl)piperazine
- 1-(2-iodobenzyl)indole-2-carbaldehyde
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1 - Skin Corr. 1B - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
235.4 °F - closed cup
Flash Point(C)
113 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
An Approach to α-substituted Amines.
Williams L, et al.
Tetrahedron, 50(48), 13697-13708 (1994)
Sushama A. Dandekar et al.
The Journal of organic chemistry, 64(5), 1543-1553 (2001-10-25)
The tricyclic title compounds, 4 and 5, were synthesized by initial alkylation of the lithium monoenolates of N,N,N',N'-tetramethylbutanediamide (6) and N,N,N',N'-tetramethylpentanediamide (19) with 2-iodobenzyl chloride in liquid NH(3) at -60 degrees C to afford 2-(2-iodobenzyl)-N,N,N',N'-tetramethylbutanediamide (9) and 2-(2-iodobenzyl)-N,N,N',N'-tetramethylpentanediamide (20) in
1-Azatriene cyclisation as a route to annelated pyrido [4, 3-b] indoles.
Gilchrist TL ,et al.
Tetrahedron, 53(12), 4447-4456 (1997)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service