All Photos(1)
About This Item
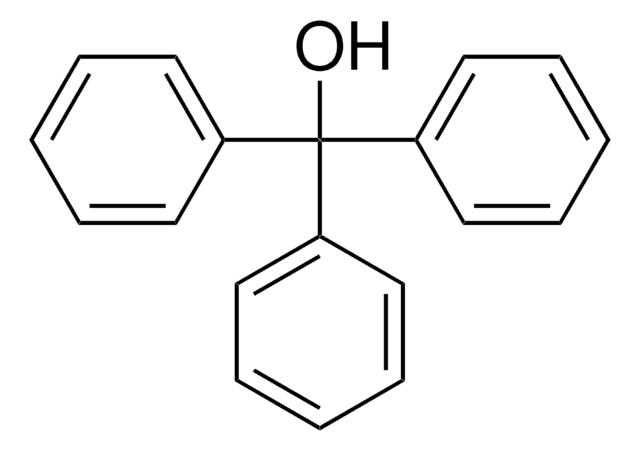
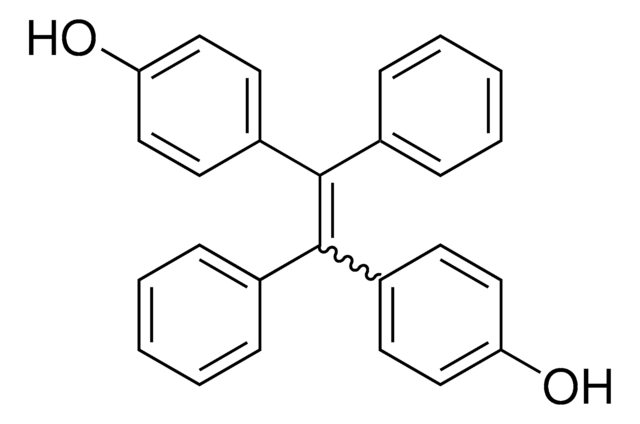
Linear Formula:
(C6H5)3CC6H4OH
CAS Number:
Molecular Weight:
336.43
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
mp
283-286 °C (lit.)
SMILES string
Oc1ccc(cc1)C(c2ccccc2)(c3ccccc3)c4ccccc4
InChI
1S/C25H20O/c26-24-18-16-23(17-19-24)25(20-10-4-1-5-11-20,21-12-6-2-7-13-21)22-14-8-3-9-15-22/h1-19,26H
InChI key
NIPKXTKKYSKEON-UHFFFAOYSA-N
Application
4-Tritylphenol was used in the synthesis of 5-arylethynyl-2′-deoxyuridines and propargyl ethers. It may be used in the synthesis of pyridyl rotaxane.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Bilge Baytekin et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 14(32), 10012-10028 (2008-10-01)
Tetralactam macrocycles can be functionalized by a variety of cross-coupling reactions. A modular "toolbox" strategy is presented that allows 1) several tetralactam macrocycles to be covalently connected with each other or with a central spacer, 2) the macrocycles to be
Mikhail V Skorobogatyi et al.
Organic & biomolecular chemistry, 4(6), 1091-1096 (2006-03-10)
Three new 5-arylethynyl-2'-deoxyuridines containing bulky aryls have been prepared and tested against HSV-1 in Vero cells. The introduction of a substituent in the phenyl group of an inactive compound, 5-phenylethynyl-2'-deoxyuridine, leads to the appearance of anti-HSV properties. The most active
Christopher Elam et al.
European journal of medicinal chemistry, 46(5), 1512-1523 (2011-03-01)
Two screening protocols based on recursive partitioning and computational ligand docking methodologies, respectively, were employed for virtual screens of a compound library with 345,000 entries for novel inhibitors of the enzyme sarco/endoplasmic reticulum calcium ATPase (SERCA), a potential target for
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service