D102504
1,5-Difluoro-2,4-dinitrobenzene
97%
Synonym(s):
DFDNB
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
F2C6H2(NO2)2
CAS Number:
Molecular Weight:
204.09
Beilstein:
1883116
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
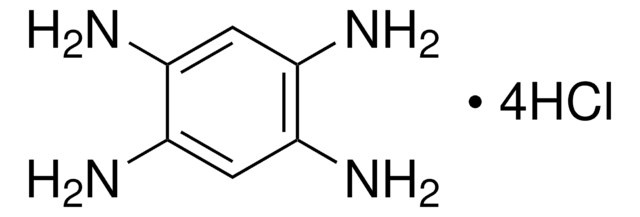
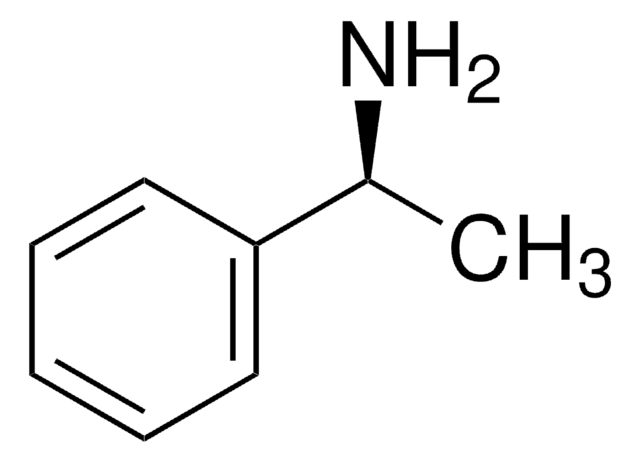
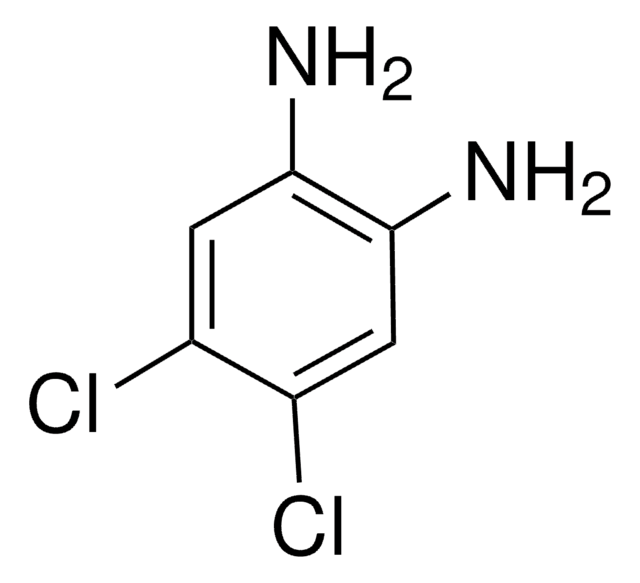
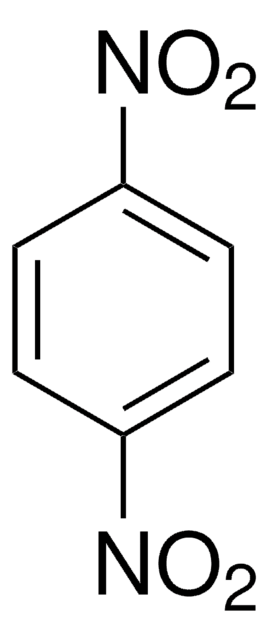
Recommended Products
Assay
97%
form
powder
mp
72-74 °C (lit.)
SMILES string
[O-][N+](=O)c1cc(c(F)cc1F)[N+]([O-])=O
InChI
1S/C6H2F2N2O4/c7-3-1-4(8)6(10(13)14)2-5(3)9(11)12/h1-2H
InChI key
VILFTWLXLYIEMV-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 2 Oral - Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - STOT RE 2
Storage Class Code
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Zhanguo Wang et al.
Journal of combinatorial chemistry, 9(4), 652-660 (2007-05-17)
This paper discusses the synthesis of privileged structures 4H-benzo[1,4]thiazin-3-one and 1,1-dioxo-1,4-dihydro-2H-1lambda6-benzo[1,4]thiazin-3-one derivatives in a parallel solution-phase manner using 1,5-difluoro-2,4-dinitrobenzene. Each scaffold possesses four diversity points. A cheap and efficient oxidant, urea-hydrogen peroxide (UHP), was applied for the introduction of the
Yunyun Yuan et al.
Journal of combinatorial chemistry, 9(1), 158-170 (2007-01-09)
This paper discusses the synthesis of benzo[1,4]oxazin-3-one-based compounds from 1,5-difluoro-2,4-dinitrobenzene (1), including benzo[1,4]oxazin-3-ones (5-11) and five novel benzo[1,4]oxazin-3-one-based tricycles: 6-hydroxy-4H-1-oxa-4,5,8-triazaanthracen-3-one (14), 3,8-dihydro-5-oxa-1,3,8-triazacyclopenta-[b]-naphthalene-7-one (15, 17, 21), 3,8-dihydro-5-oxa-1,2,3,8-tetraazacylopenta[b]-naphthalene-7-one (16, 20), 3,8-dihydro-1H-5-oxa-1,3,8-triazacyclopenta[b]-naphthalene-2,7-dione (18, 22), and 5,8-dihydro-4H-1-oxa-4,5,8-triazaanthracene-3,6,7-trione (19). Finally, a chemical library based on
S Simonsson et al.
The Journal of biological chemistry, 273(38), 24633-24639 (1998-09-12)
The UL9 gene of herpes simplex virus type 1 (HSV-1) encodes an origin binding protein (OBP). It is an ATP-dependent DNA helicase and a sequence-specific DNA-binding protein. The latter function is carried out by the C-terminal domain of OBP (DeltaOBP).
Yanshun Liu et al.
Protein science : a publication of the Protein Society, 11(2), 371-380 (2002-01-16)
When concentrated in mildly acidic solutions, bovine pancreatic ribonuclease (RNase A) forms long-lived oligomers including two types of dimer, two types of trimer, and higher oligomers. In previous crystallographic work, we found that the major dimeric component forms by a
Alexander Shivanyuk et al.
Organic letters, 4(9), 1555-1558 (2002-04-27)
[reaction: see text]. o-alkylation of C2V-symmetrical resorcinarene tetraesters 2 with 2 equiv of 1,3-difluoro-4,6-dinitrobenzene readily affords conformationally rigid octanitro resorcinarene 3, which is a potential scaffold for the design of supramolecular structures.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service


![1,8-Diazabicyclo[5.4.0]undec-7-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)