69723
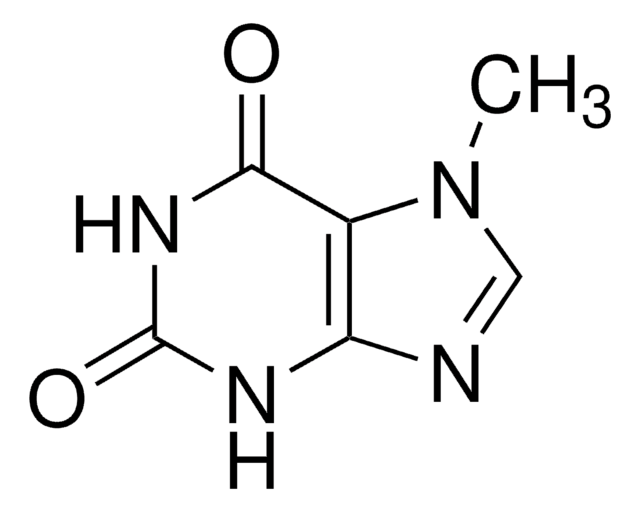
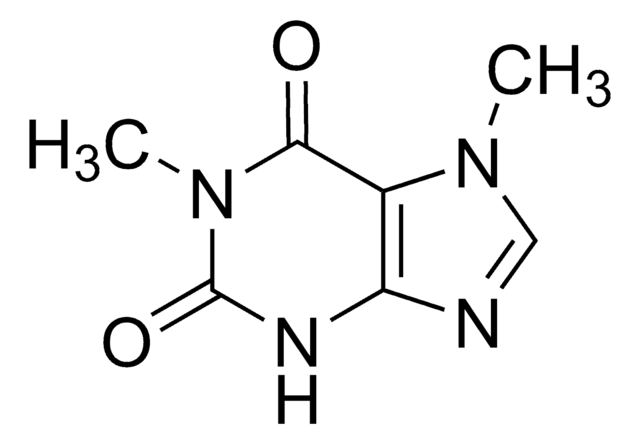
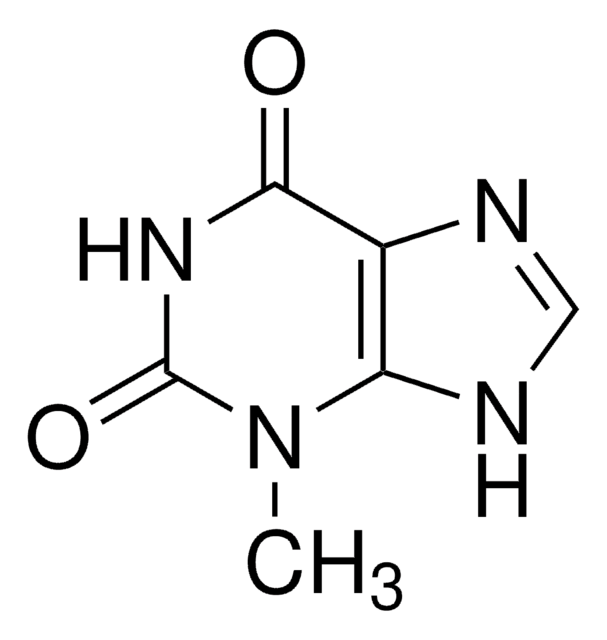
7-Methylxanthine
≥98.0% (HPLC)
Synonym(s):
2,6-Dihydroxy-7-methylpurine, Heteroxanthine
About This Item
Recommended Products
Assay
≥98.0% (HPLC)
form
powder
mp
≥300 °C
SMILES string
Cn1cnc2NC(=O)NC(=O)c12
InChI
1S/C6H6N4O2/c1-10-2-7-4-3(10)5(11)9-6(12)8-4/h2H,1H3,(H2,8,9,11,12)
InChI key
PFWLFWPASULGAN-UHFFFAOYSA-N
Gene Information
rat ... Adora1(29290) , Adora2a(25369)
Looking for similar products? Visit Product Comparison Guide
General description
Application
Packaging
related product
Storage Class Code
13 - Non Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
Xanthine is a purine base found in most human body tissues and fluids as well as in other organisms. Methylated xanthines (methylxanthines), which include caffeine, paraxanthine, theobromine, and theophylline, commonly used for their effects as mild stiµlants and as bronchodilators, notably in the treatment of asthma symptoms. This application shows the efficient separation of several common xanthines and may be applied their analysis in any number of desired matrices.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service