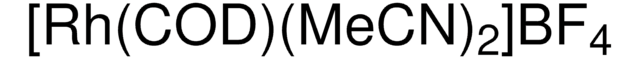683175
Chiralyst P374
Umicore
Synonym(s):
Bis(norbornadiene)rhodium(I) tetrafluoroborate, Bis(bicyclo[2.2.1]hepta-2,5-diene)rhodium tetrafluoroborate, Bis(norbornadiene)(tetrafluoroborato)rhodium, Rh(nbd)2BF4, Rh(nbd)2BF4
About This Item
Recommended Products
Quality Level
form
solid
reaction suitability
core: rhodium
reagent type: catalyst
mp
136.9-139.9 °C
SMILES string
[Rh+].F[B-](F)(F)F.C1C2C=CC1C=C2.C3C4C=CC3C=C4
InChI
1S/2C7H8.BF4.Rh/c2*1-2-7-4-3-6(1)5-7;2-1(3,4)5;/h2*1-4,6-7H,5H2;;/q;;-1;+1/t2*6-,7+;;
InChI key
HAYDJWBQWOEERB-JAGGYEKFSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- Lactones via the 1,4-addition of arylboronic acids to butenolide.
- Chiral β-aryl-substituted ketones and esters.
- β-aryloxycarboxylic esters from β-aryloxy-α,γ-unsaturated esters.
Legal Information
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1 - Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
We are happy to offer a series of precatalysts for asymmetric catalysis and Pd based cross coupling catalysis. Our partnership with Umicore allows us to offer a portfolio of metal complexes with batch-to-batch consistency for a plethora of catalytic reactions.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service![(R)-3,3′-Bis[3,5-bis(trifluoromethyl)phenyl]-1,1′-binaphthyl-2,2′-diyl hydrogenphosphate 95%](/deepweb/assets/sigmaaldrich/product/structures/270/636/14dc9413-bcb4-478c-8e4d-3605317c13a5/640/14dc9413-bcb4-478c-8e4d-3605317c13a5.png)
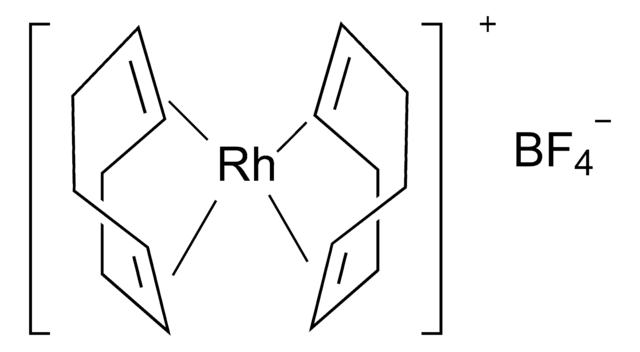


![Bis(1,5-cyclooctadiene)rhodium(I) tetrakis[bis(3,5-trifluoromethyl)phenyl]borate](/deepweb/assets/sigmaaldrich/product/structures/275/399/3f196135-3b90-4f6f-95b0-512766f1f5e1/640/3f196135-3b90-4f6f-95b0-512766f1f5e1.png)
![(S,R,R)-(+)-(3,5-Dioxa-4-phosphacyclohepta[2,1-a:3,4-a′]dinaphthalen-4-yl)bis(1-phenylethyl)amine 95% (HPLC)](/deepweb/assets/sigmaaldrich/product/structures/366/790/7555ef31-5d0b-45c9-ad40-5dfd0fe85125/640/7555ef31-5d0b-45c9-ad40-5dfd0fe85125.png)