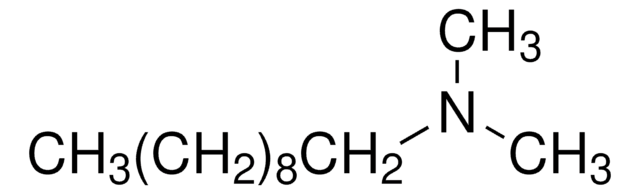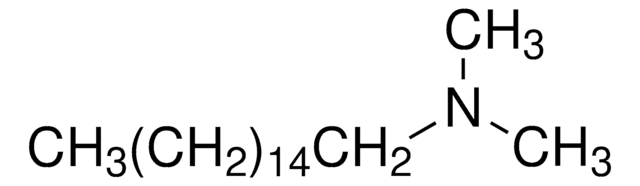538639
N,N-Dimethyldecylamine
≥90%
Synonym(s):
1-(Dimethylamino)decane
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
CH3(CH2)9N(CH3)2
CAS Number:
Molecular Weight:
185.35
Beilstein:
1738198
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
≥90%
refractive index
n20/D 1.431 (lit.)
bp
234 °C (lit.)
density
0.778 g/mL at 25 °C (lit.)
functional group
amine
SMILES string
CCCCCCCCCCN(C)C
InChI
1S/C12H27N/c1-4-5-6-7-8-9-10-11-12-13(2)3/h4-12H2,1-3H3
InChI key
YWWNNLPSZSEZNZ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
N,N-Dimethyldecylamine is a tertiary amine. It can be prepared by reacting N,N-dimethyldodecylamine, t-butyl hydroperoxide, vanadium oxyacetylacetonate and t-butyl alcohol.
Application
N,N-Dimethyldecylamine may be used as one of the reaction components in the preparation of:
- comb-shaped quaternized poly(2,6-dimethylphenylene oxide) (PPO)
- pore-expanded MCM-41 silica
- QA C10 stationary phase bearing quaternary ammonium embedded between a propyl and a decyl chain
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Skin Irrit. 2
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
197.6 °F - closed cup
Flash Point(C)
92 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Hydroperoxide oxidations catalyzed by metals. II. Oxidation of tertiary amines to amine oxides.
Sheng MN and Zajacek JG.
The Journal of Organic Chemistry, 33(2), 588-590 (1968)
Applications of pore-expanded MCM-41 silica: 4. Synthesis of a highly active base catalyst.
Das DD, et al.
Catalysis Communications, 8(5), 829-833 (2007)
Nanwen Li et al.
Journal of the American Chemical Society, 135(27), 10124-10133 (2013-06-01)
To produce an anion-conductive and durable polymer electrolyte for alkaline fuel cell applications, a series of quaternized poly(2,6-dimethyl phenylene oxide)s containing long alkyl side chains pendant to the nitrogen-centered cation were synthesized using a Menshutkin reaction to form comb-shaped structures.
Qiaoxia Liu et al.
Journal of separation science, 35(20), 2685-2692 (2012-09-26)
A stationary phase (named QA C10) with quaternary ammonium embedded between a propyl and a decyl chain was synthesized by immobilization of N,N-dimethyldecylamine on chloropropyl-silica surface. A set of representative neutral, basic, and acidic compounds was employed to evaluate its
Daria Szymaniak et al.
ChemPlusChem, 85(10), 2281-2289 (2020-09-23)
The synthesis and characteristics are presented of novel double-salt herbicidal ionic liquids (DSHILs) that contain 4-chloro-2-methylphenoxyacetate and trans-cinnamate anions. In the designed synthesis, an anion of natural origin and a herbicidal anion were combined with an amphiphilic bisammonium cation to
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service