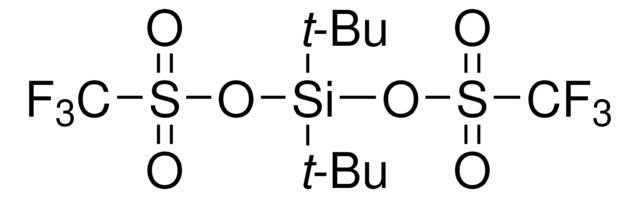337005
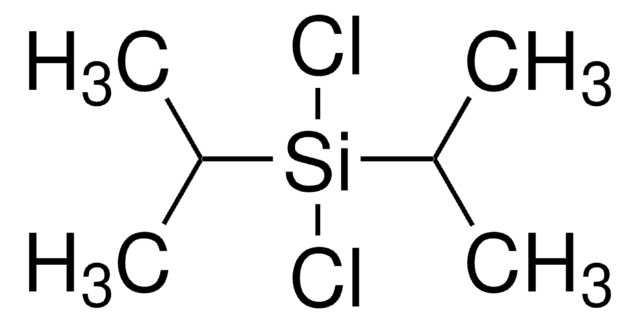
1,3-Dichloro-1,1,3,3-tetraisopropyldisiloxane
97%
Synonym(s):
TIPDSiCl2
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
[(CH3)2CH]2Si(Cl)OSi(Cl)[CH(CH3)2]2
CAS Number:
Molecular Weight:
315.43
Beilstein:
1934216
MDL number:
UNSPSC Code:
12352001
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
form
liquid
refractive index
n20/D 1.454 (lit.)
bp
70 °C/0.5 mmHg (lit.)
density
0.986 g/mL at 25 °C (lit.)
SMILES string
CC(C)[Si](Cl)(O[Si](Cl)(C(C)C)C(C)C)C(C)C
InChI
1S/C12H28Cl2OSi2/c1-9(2)16(13,10(3)4)15-17(14,11(5)6)12(7)8/h9-12H,1-8H3
InChI key
DDYAZDRFUVZBMM-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1 - Met. Corr. 1 - Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
168.8 °F
Flash Point(C)
76 °C
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Aldrichimica Acta, 15, 11-11 (1982)
Suetying Chow et al.
Nucleosides, nucleotides & nucleic acids, 22(5-8), 583-587 (2003-10-21)
An improved strategy for the synthesis of 2'-O-methyl-guanosine (6) and 2'-MOE-guanosine (8) is reported. The regioselectivity of the alkylation was attained using a novel silicon-based protecting group, methylene-bis (diisopropyl-silylchloride) (MDPSCl2, 2). The alkylation proceeded in a chemoselective manner using NaHMDS
M Kwiatkowski et al.
Nucleic acids research, 24(23), 4632-4638 (1996-12-01)
The synthesis of oligodeoxynucleotides is marred by several problems that contribute to the formation of defective molecules. This in turn seriously limits the usefulness of such reagents in DNA diagnostics, molecular cloning, DNA structural analysis and in antisense therapy. In
The Journal of Organic Chemistry, 58, 2552-2552 (1993)
W T Markiewicz et al.
Nucleic acids symposium series, (7)(7), 115-127 (1980-01-01)
Recent results concerning the synthesis of oligoribonucleotides via the phosphotriester method, such as functionalization of ribonucleosides, new phosphorylating agents, 5'-O-sulfonylation and chromatography on Sephadex LH-20 for monitoring the removal of internucleotide phosphotriester groups, are presented. To show that efficiency of
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service