289345
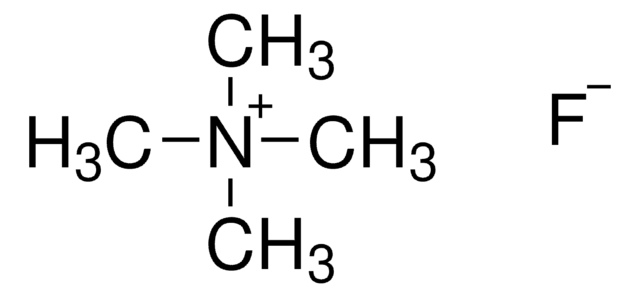
Cesium fluoride
99.9% trace metals basis
Synonym(s):
NSC 84270
Sign Into View Organizational & Contract Pricing
All Photos(5)
About This Item
Empirical Formula (Hill Notation):
CsF
CAS Number:
Molecular Weight:
151.90
EC Number:
MDL number:
UNSPSC Code:
12352302
PubChem Substance ID:
NACRES:
NA.22
grade:
for analytical purposes
Recommended Products
grade
for analytical purposes
Quality Level
Assay
99.9% trace metals basis
mp
682 °C (lit.)
density
4.115 g/mL at 25 °C (lit.)
SMILES string
[F-].[Cs+]
InChI
1S/Cs.FH/h;1H/q+1;/p-1
InChI key
XJHCXCQVJFPJIK-UHFFFAOYSA-M
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
Reactant for:
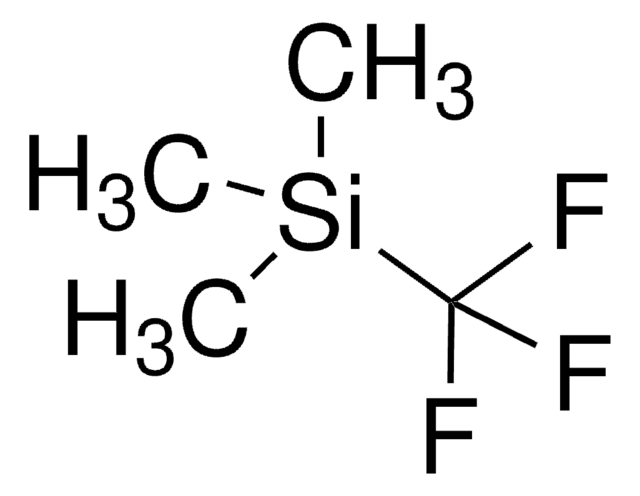
- Preparation of building blocks for synthesis of fluoroallylic compounds
- Synthesis of alcohols via hydrolysis of alkyl silyl ethers at neutral pH in buffered mixed organic-aqueous solutions
- Nucleophilic fluorination of alkynyliodonium salts to form fluorovinylic compounds
- Nucleophilic aromatic substitution (SNAr) reactions
Used as a base in a Suzuki cross-coupling synthesis of ortho-substituted biaryls. Also employed as a reagent for nucleophilic fluorination of primary halides and sulfonates in protic media such as tert-butyl and tert-pentyl alcohols.
Used in the successful synthesis of a single-crystal Dion-Jacobson phase, CsLaTa2O7, that has applications in photocatalysis and superconductivity.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Chronic 3 - Eye Dam. 1 - Repr. 2 - STOT RE 2
Target Organs
Kidney,Adrenal gland
Supplementary Hazards
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Dong Wook Kim et al.
Journal of the American Chemical Society, 128(50), 16394-16397 (2006-12-15)
Aprotic solvents are usually preferred for the SN2 reactions, because nucleophilicity and hence SN2 reactivity are severely retarded by the influence of the partial positive charge of protic solvents. In this work, we introduce a remarkable effect of using tertiary
Journal of Organometallic Chemistry, 691, 5688-5688 (2006)
Takashi Okitsu et al.
Chemical communications (Cambridge, England), (47)(47), 6330-6332 (2008-12-03)
A highly efficient and rapid total synthesis of 9Z-retinoic acid was accomplished by caesium fluoride-promoted Stille coupling reaction; using a common building block, 9Z-retinoic acid analogues were also prepared by the same method without isomerisation of the Z-double bond.
S Nelson et al.
Science (New York, N.Y.), 265(5173), 774-777 (1994-08-05)
Neurons in the primary visual cortex of the cat are selectively activated by stimuli with particular orientations. This selectivity can be disrupted by the application of antagonists of the inhibitory neurotransmitter gamma-aminobutyric acid (GABA) to a local region of the
Nongmaithem Jiten Singh et al.
The journal of physical chemistry. B, 110(8), 3808-3815 (2006-02-24)
The structures, stabilities, thermodynamic quantities, dissociation energies, infrared spectra, and electronic properties of CsF hydrated by water molecules are investigated by using density functional theory, Møller-Plesset second-order perturbation theory (MP2), coupled cluster theory with singles, doubles, and perturbative triples excitations
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 289345-5G | 4061826284223 |
| 289345-100G | 4061826284216 |
| 289345-1KG | 4061826732328 |
| 289345-25G | 4061833545980 |
| 289345-500G | 4061833545997 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service