17324
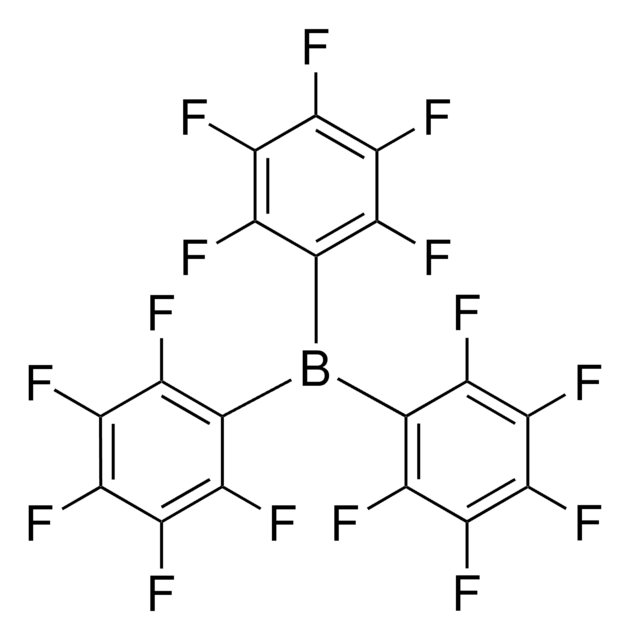
Lithium tetrakis(pentafluorophenyl)borate ethyl etherate
Synonym(s):
Tetrakis(pentafluorophenyl)boron lithium ethyl etherate
About This Item
Recommended Products
reaction suitability
core: boron
reagent type: catalyst
Quality Level
mp
117-122 °C
SMILES string
[Li+].CCOCC.Fc1c(F)c(F)c(c(F)c1F)[B-](c2c(F)c(F)c(F)c(F)c2F)(c3c(F)c(F)c(F)c(F)c3F)c4c(F)c(F)c(F)c(F)c4F
InChI
1S/C24BF20.C4H10O.Li/c26-5-1(6(27)14(35)21(42)13(5)34)25(2-7(28)15(36)22(43)16(37)8(2)29,3-9(30)17(38)23(44)18(39)10(3)31)4-11(32)19(40)24(45)20(41)12(4)33;1-3-5-4-2;/h;3-4H2,1-2H3;/q-1;;+1
InChI key
KPLZKJQZPFREPG-UHFFFAOYSA-N
Application
- A coordinating counter anion in electrochemical reactions along with transition metal catalysts to enhance their acidity or solubility.
- A catalyst in the Baeyer-Villiger oxidation of cycloalkanones to obtain lactones in the presence of aqueous hydrogen peroxide and oxalic acid.
- An activator in the synthesis of poly(norbornene ester)s.
Packaging
Other Notes
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
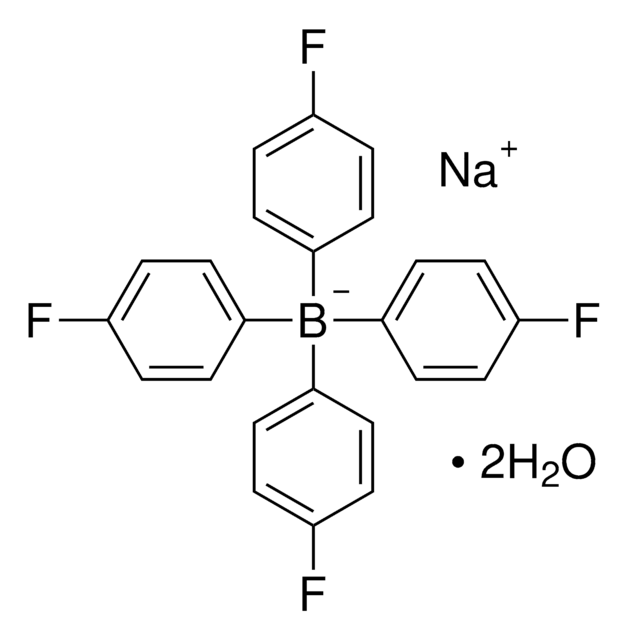
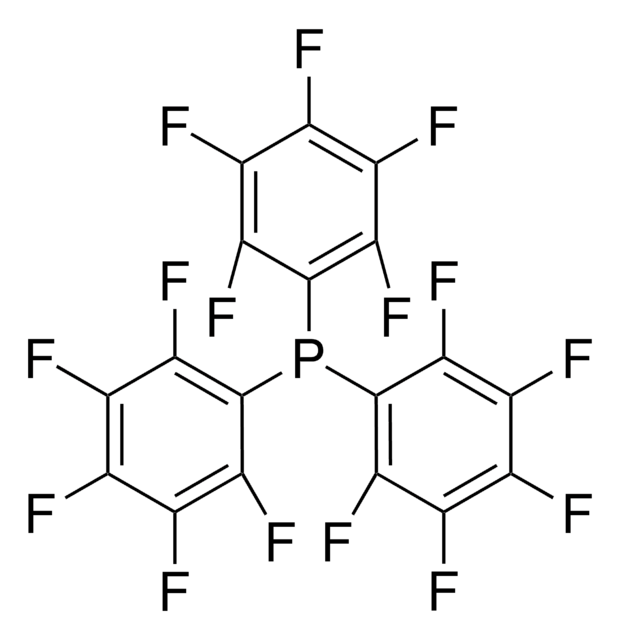
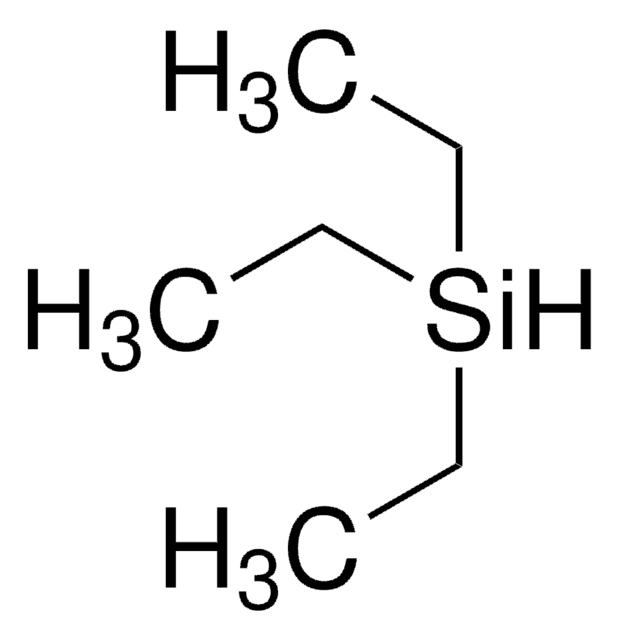
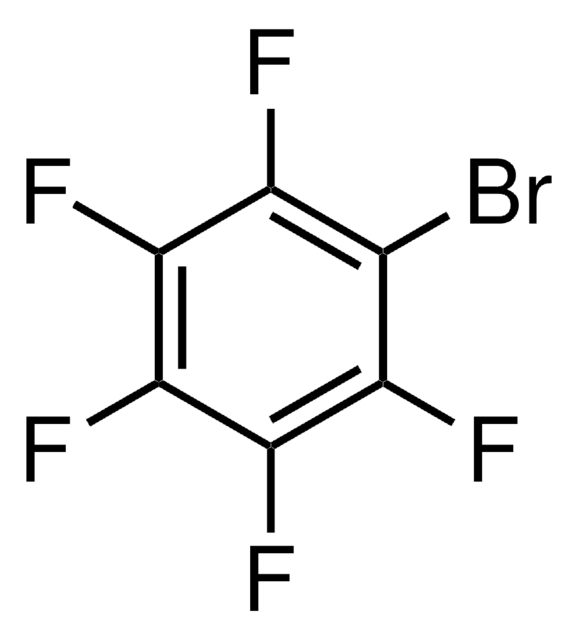
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service![Sodium tetrakis[3,5-bis(trifluoromethyl)phenyl]borate](/deepweb/assets/sigmaaldrich/product/structures/251/439/7a621e74-bfd1-4a43-833c-09adfcc1e0b3/640/7a621e74-bfd1-4a43-833c-09adfcc1e0b3.png)
![Potassium tetrakis[3,5-bis(trifluoromethyl)phenyl]borate Selectophore™](/deepweb/assets/sigmaaldrich/product/structures/631/130/b5486f44-2e69-40d0-902f-dd71894a6add/640/b5486f44-2e69-40d0-902f-dd71894a6add.png)