138622
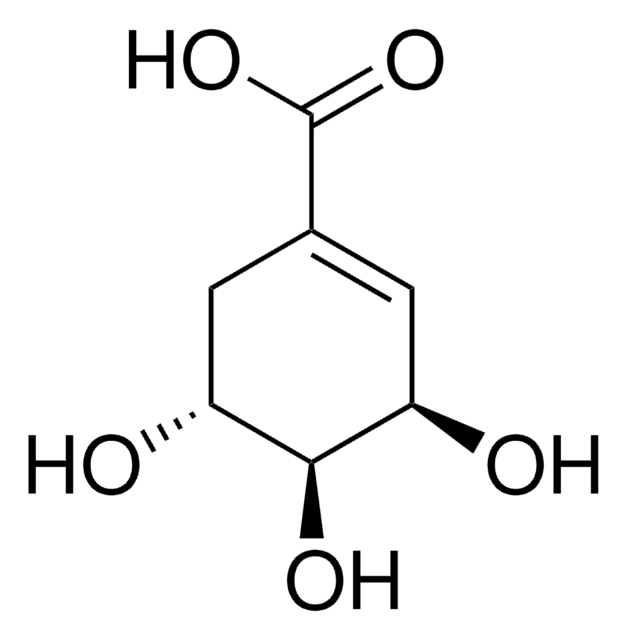
D-(−)-Quinic acid
98%
Synonym(s):
(-)-Quinic acid, (1alpha,3R,4alpha,5R)-1,3,4,5-Tetrahydroxycyclohexanecarboxylic acid, D-(-)-Quinic acid
About This Item
Recommended Products
Quality Level
Assay
98%
form
powder
optical activity
[α]20/D −43.9°, c = 11.2 in H2O
functional group
carboxylic acid
hydroxyl
SMILES string
O[C@@H]1C[C@@](O)(C[C@@H](O)[C@H]1O)C(O)=O
InChI
1S/C7H12O6/c8-3-1-7(13,6(11)12)2-4(9)5(3)10/h3-5,8-10,13H,1-2H2,(H,11,12)/t3-,4-,5-,7+/m1/s1
InChI key
AAWZDTNXLSGCEK-WYWMIBKRSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- A chiral selector electrolyte along with copper(II) sulfate. This electrolyte is utilized in chiral resolution DL-tartaric acid by ligand-exchange capillary electrophoresis method.
- A starting material in the synthesis of stereoisomers of 3,4,6-trihydroxyazepanes, 7-hydroxymethyl-3,4,5-trihydroxyazepanes, and 3,4,5-trihydroxyazepanes, as potential inhibitors of glycosidase.
- A precursor for the preparation of trihydroxy piperidine derivatives and (+)-proto-quercitol glycosidase inhibitors.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 138622-25G | 4061838732408 |
| 138622-100G | 4061838732392 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service