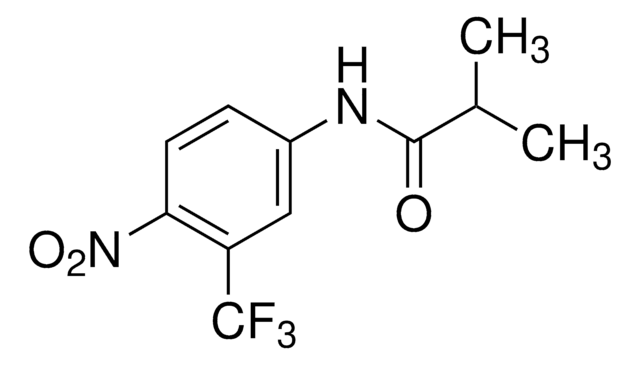
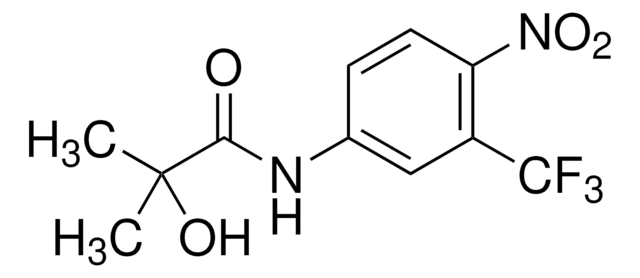
推薦產品
應用
Nilutamide has been used:
- as a nuclear androgen receptor (nAR) inhibitor to study its effects on oocyte maturation in zebrafish
- to determine its effects on bioluminescent Saccharomyces cerevisiae bioreporters in BLYAS assay
- as a substrate in hydrogenation reaction
生化/生理作用
Nilutamide is a nuclear androgen receptor inhibitor. It is a nonsteroidal antiandrogen that competitively inhibits the binding of dihydrotestosterone to the androgen receptor. Nilutamide shows a therapeutic effect against prostate cancer.
特點和優勢
This compound is featured on the Nuclear Receptors (Steroids) page of the Handbook of Receptor Classification and Signal Transduction. To browse other handbook pages, click here.
This compound was developed by Sanofi Aventis. To browse the list of other pharma-developed compounds and Approved Drugs/Drug Candidates, click here.
訊號詞
Danger
危險聲明
危險分類
Acute Tox. 3 Oral - Repr. 1B
儲存類別代碼
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
分析證明 (COA)
輸入產品批次/批號來搜索 分析證明 (COA)。在產品’s標籤上找到批次和批號,寫有 ‘Lot’或‘Batch’.。
A Desai et al.
Urology, 58(6), 1016-1020 (2001-12-18)
To study the ability of nilutamide to induce prostate-specific antigen (PSA) responses in patients with hormone-resistant prostate cancer who had been exposed to prior antiandrogen therapy, because resistance to antiandrogens may be mediated by altered binding to a mutated or
Ravi A Madan et al.
Clinical cancer research : an official journal of the American Association for Cancer Research, 14(14), 4526-4531 (2008-07-17)
We reported previously the first randomized study of any kind in patients with nonmetastatic, castrate-resistant prostate cancer. The study employed vaccine, the hormone nilutamide, and the combined therapy (crossover for each arm) with an endpoint of time to progression. We
Richard Choo et al.
International journal of radiation oncology, biology, physics, 75(4), 983-989 (2009-05-05)
To determine the efficacy of a combined approach of radiotherapy (RT) plus 2-year androgen suppression (AS) as salvage treatment for prostate-specific antigen (PSA) relapse after radical prostatectomy (RP). Seventy-five patients with PSA relapse after RP were treated with salvage RT
Masayasu Urushibara et al.
The Prostate, 67(8), 799-807 (2007-03-22)
Molecular basis for secondary antiandrogen therapy in prostate cancer with mutant androgen receptors (ARs) is not fully elucidated. Effects of steroidal and non-steroidal antiandrogens on transcriptional activities of wild-type and mutant (W741C, T877A, and W741C+T877A) ARs were measured. Crystal structure
Jennifer Keiser et al.
The Journal of antimicrobial chemotherapy, 65(9), 1991-1995 (2010-06-26)
The antischistosomal properties of the marketed antiandrogens bicalutamide, flutamide, nilutamide and cyproterone acetate were studied both in vivo and in vitro. Schistosoma mansoni-infected mice were treated orally with 50-400 mg/kg of the antiandrogens 3 and 7 weeks post-infection. In addition
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務