推薦產品
product name
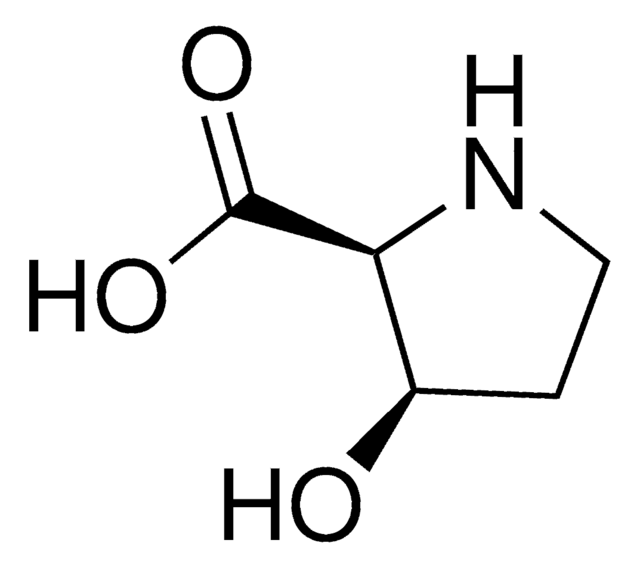
cis-4-Hydroxy-D-proline,
化驗
≥98% (TLC)
形狀
powder
技術
ligand binding assay: suitable
顏色
white
mp
243 °C (dec.) (lit.)
應用
peptide synthesis
SMILES 字串
O[C@H]1CN[C@H](C1)C(O)=O
InChI
1S/C5H9NO3/c7-3-1-4(5(8)9)6-2-3/h3-4,6-7H,1-2H2,(H,8,9)/t3-,4-/m1/s1
InChI 密鑰
PMMYEEVYMWASQN-QWWZWVQMSA-N
尋找類似的產品? 前往 產品比較指南
相關類別
生化/生理作用
Cis-4-Hydroxy-D-proline may be used as a starting material for the 13-step synthesis of new conformationally restricted PNA adenine monomer and the synthesis of N-Benzyl pyrrolidinyl sordaricin derivatives. Cis-4-Hydroxy-D-proline is a substrate that may be used to study the specificity and kinetics of D-alanine dehydrogenase. Cis-4-Hydroxy-D-proline may be used to analyze the substrate specificity of amino acid transporter PAT1.
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Gloves, type N95 (US)
分析證明 (COA)
輸入產品批次/批號來搜索 分析證明 (COA)。在產品’s標籤上找到批次和批號,寫有 ‘Lot’或‘Batch’.。
客戶也查看了
A Püschl et al.
Organic letters, 2(26), 4161-4163 (2001-01-11)
[reaction:see text] A new conformationally restricted PNA adenine monomer has been synthesized in 13 steps from cis-4-hydroxy-D-proline. A fully modified adenine decamer displayed improved binding affinity toward complementary DNA and RNA oligonucleotides as compared to that of the parent PNA
Masami Arai et al.
Bioorganic & medicinal chemistry letters, 12(19), 2733-2736 (2002-09-10)
N-Benzyl pyrrolidinyl sordaricin derivatives have been synthesized from cis-4-hydroxy-D-proline in a stereocontrolled manner. These compounds maintained moderate antifungal activity against several pathogenic fungal strains. Their MIC values against Candida albicans were in the range of 0.25-2 microg/mL.
L Metzner et al.
Amino acids, 31(2), 111-117 (2006-05-16)
The proton coupled amino acid transporter PAT1 expressed in intestine, brain, and other organs accepts L- and D-proline, glycine, and L-alanine but also pharmaceutically active amino acid derivatives such as 3-amino-1-propanesulfonic acid, L-azetidine-2-carboxylic acid, and cis-4-hydroxy-D-proline as substrates. We systematically
Charles E Deutch
FEMS microbiology letters, 238(2), 383-389 (2004-09-11)
3,4-Dehydro-DL-proline is a toxic analogue of L-proline which has been useful in studying the uptake and metabolism of this key amino acid. When membrane fractions from Escherichia coli strain UMM5 (putA1::Tn5 proC24) lacking both L-proline dehydrogenase and L-Delta(1)-pyrroline-5-carboxylate reductase were
Ashley C Campbell et al.
Archives of biochemistry and biophysics, 698, 108727-108727 (2020-12-18)
Proline utilization A (PutA) proteins are bifunctional proline catabolic enzymes that catalyze the 4-electron oxidation of l-proline to l-glutamate using spatially-separated proline dehydrogenase and l-glutamate-γ-semialdehyde dehydrogenase (GSALDH, a.k.a. ALDH4A1) active sites. The observation that l-proline inhibits both the GSALDH activity
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務