全部照片(3)
About This Item
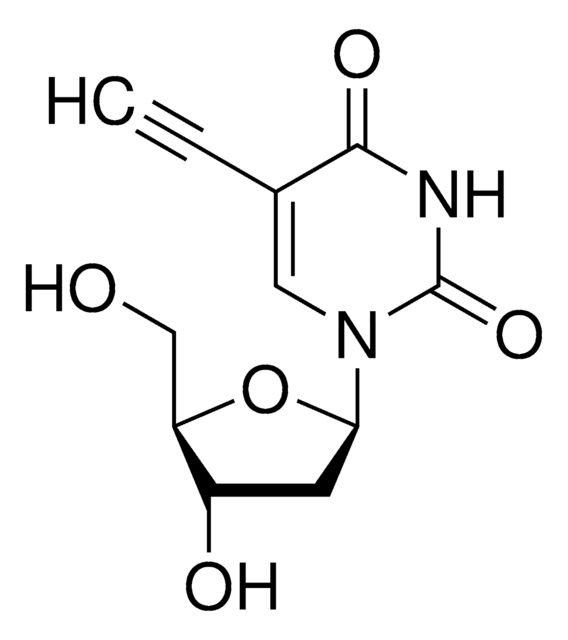
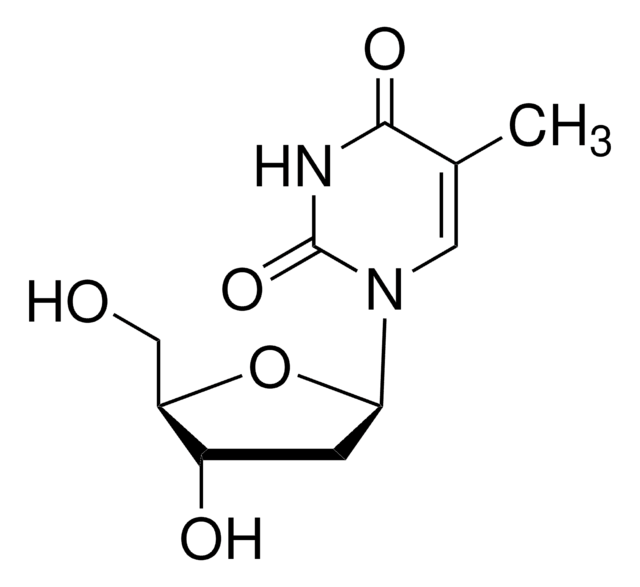
經驗公式(希爾表示法):
C11H16N2O5
CAS號碼:
分子量::
256.26
EC號碼:
MDL號碼:
分類程式碼代碼:
41106305
PubChem物質ID:
NACRES:
NA.51
推薦產品
生物源
synthetic (organic)
品質等級
化驗
≥95% (HPLC)
形狀
powder
溶解度
water: 50 mg/mL, clear, colorless to yellow
儲存溫度
−20°C
SMILES 字串
CCC1=CN(C2CC(O)C(CO)O2)C(=O)NC1=O
InChI
1S/C11H16N2O5/c1-2-6-4-13(11(17)12-10(6)16)9-3-7(15)8(5-14)18-9/h4,7-9,14-15H,2-3,5H2,1H3,(H,12,16,17)
InChI 密鑰
XACKNLSZYYIACO-UHFFFAOYSA-N
生化/生理作用
5-Ethyl-2′-deoxyuridine (EUdR) is used as a 5-fluorouracil (FU) modulator. EtdUrd may be used to enhance the therapeutic index of 5-FU by reducing the catabolism, prolonging the plasma and intratumoral concentrations of 5-FU, and offering protection to normal organs by increasing the endogenous uridine levels.
5-Ethyl-2′-deoxyuridine is an analog of pyrimidine, uridine. It is an effective antiviral agent and inhibits cell division in leukemic tumor cells. 5-Ethyl-2′-deoxyuridine interacts with 2′-deoxycytidine phosphate (dCTP) biosynthesis pathway to elicit antiproliferative effects.
聯結
Homolog of thymidine
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Gloves, type N95 (US)
客戶也查看了
E De Clercq et al.
The Journal of biological chemistry, 262(31), 14905-14911 (1987-11-05)
5-Ethyl-2'-deoxyuridine (EDU) is a potent and selective inhibitor of the replication of herpes simplex virus type 1 (HSV-1) and 2 (HSV-2), which is currently being pursued for the topical treatment of HSV-1 and HSV-2 infections in humans. Using [4-14C]EDU as
P S Imperia et al.
Antiviral research, 9(4), 263-272 (1988-07-01)
Using an in vitro system we measured the corneal epithelial cytotoxicity and the antiviral activity of the antiviral agents idoxuridine (IDU), trifluridine (TFT), ethyldeoxyuridine (EDU), and (E)-5-(2-Bromovinyl)-2'-deoxyuridine (BVDU). Confluent rabbit corneal epithelial cell cultures were established, and the antiviral agents
Modulation of 5-fluorouracil by 5-ethyl-2'-deoxyuridine on cell lines expressing different dihydropyrimidine dehydrogenase activities.
Katona C, Timar F, Jeney A, et al.
Anti-Cancer Drug Design, 10, 561-567 (1999)
A M Cheraghali et al.
Drug metabolism and disposition: the biological fate of chemicals, 23(2), 223-226 (1995-02-01)
The pharmacokinetics and oral (po) bioavailability of 5-ethyl-2'-deoxyuridine (EDU) and its novel 5,6-dihydro prodrugs (+)-trans-(5R,6R)-5-bromo-5-ethyl-6-ethoxy-5,6-dihydro-2'-deoxyuridine (BEEDU) and (+)-trans-(5R,6R)-5-bromo-5-ethyl-6-ethoxy-5,6-dihydro-5'-O-valeryl-2'- deoxyuridine (VBEEDU) were determined in male Balb/C mice following intravenous and no administration of a 0.4 mmol/kg dose. EDU was eliminated from
Synthesis and antiviral activity of acyclic derivatives of 5-ethyl-2'-deoxyuridine.
K Keppeler et al.
Archiv der Pharmazie, 319(4), 360-365 (1986-04-01)
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務