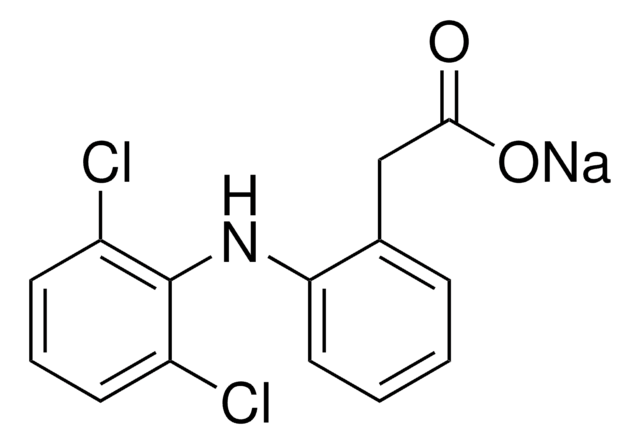
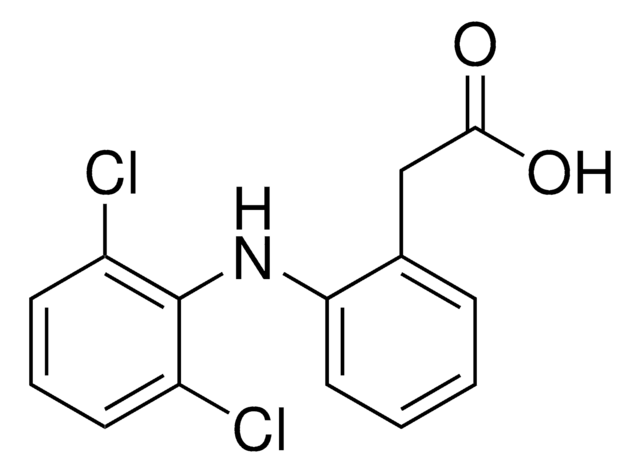
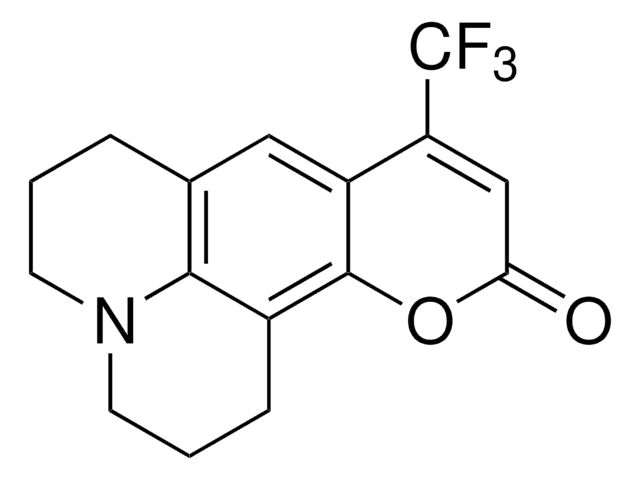
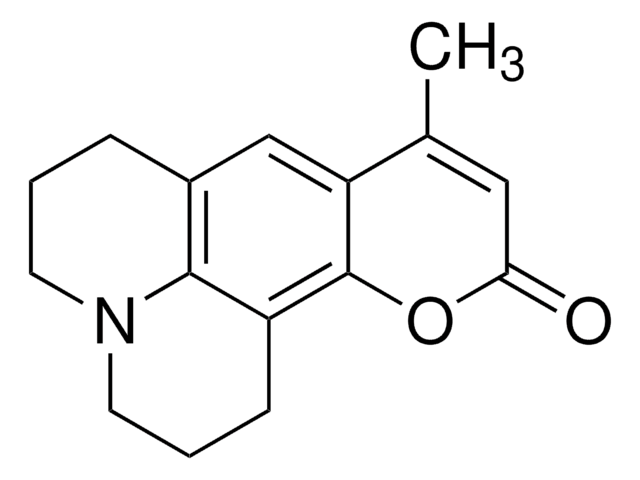
推薦產品
蒸汽壓力
0.01 mmHg ( 47 °C)
化驗
≥99% (HPLC)
形狀
powder
bp
298 °C (lit.)
mp
68-73 °C (lit.)
SMILES 字串
O=C1Oc2ccccc2C=C1
InChI
1S/C9H6O2/c10-9-6-5-7-3-1-2-4-8(7)11-9/h1-6H
InChI 密鑰
ZYGHJZDHTFUPRJ-UHFFFAOYSA-N
基因資訊
rat ... Maoa(29253) , Maob(25750)
尋找類似的產品? 前往 產品比較指南
應用
香豆素可用作制备或合成基于香豆素的抗凝血剂、抗炎剂和抗氧化剂超氧化物清除剂的前体分子。
訊號詞
Danger
危險聲明
危險分類
Acute Tox. 3 Dermal - Acute Tox. 3 Oral - Skin Sens. 1
儲存類別代碼
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
水污染物質分類(WGK)
WGK 1
閃點(°F)
323.6 °F - closed cup
閃點(°C)
162 °C - closed cup
個人防護裝備
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
客戶也查看了
Klaus Abraham et al.
Molecular nutrition & food research, 54(2), 228-239 (2009-12-22)
Coumarin is a secondary phytochemical with hepatotoxic and carcinogenic properties. For the carcinogenic effect, a genotoxic mechanism was considered possible, but was discounted by the European Food Safety Authority in 2004 based on new evidence. This allowed the derivation of
Donglei Yu et al.
Medicinal research reviews, 23(3), 322-345 (2003-03-21)
Numerous plant-derived compounds have been evaluated for inhibitory effects against HIV replication, and some coumarins have been found to inhibit different stages in the HIV replication cycle. This review article describes recent progress in the discovery, structure modification, and structure-activity
Shota Morimoto et al.
Chemical communications (Cambridge, England), 49(18), 1811-1813 (2013-01-26)
A photo-switchable fluorescent flagging approach has been developed to identify photoaffinity-labeled peptides in target protein. Upon photochemical release of the ligand, the protein was newly modified with a coumarin in place of the previously attached biotin. It allowed us to
María E Riveiro et al.
Bioorganic & medicinal chemistry, 17(18), 6547-6559 (2009-09-01)
The presumption that some coumarins might be lead compounds in the search for new differentiation agents against leukemia is based on the fact that natural coumarins, 5-(3-methyl-2-butenyloxy)-6,7-methylenedioxycoumarin (C-2) and 5-methoxy-6,7-methylenedioxycoumarin (C-1) inhibit proliferation and induce differentiation in U-937 cells [Riveiro
A Marjolein Schrijver et al.
Journal of endovascular therapy : an official journal of the International Society of Endovascular Specialists, 22(1), 87-95 (2015-03-17)
To report the results of the Dutch randomized trial comparing standard catheter-directed and ultrasound-accelerated thrombolysis (UST) for the treatment of arterial thromboembolic occlusions. The DUET study ( controlled-trials.com ; identifier ISRCTN72676102) was designed to assess whether UST can reduce therapy
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務