推薦產品
品質等級
相容性
for use with Milliflex® Rapid Systems.
一般說明
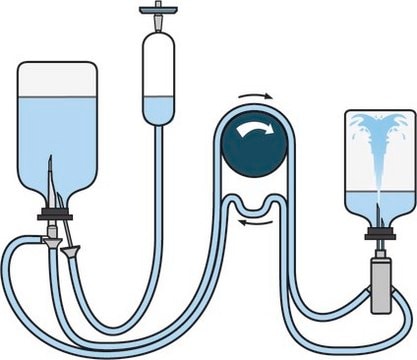
Rapid Sterility Testing Validation Protocol is designed as a ready to use step by step guide to perform PQ activities of rapid sterility testing using Milliflex® Rapid system. The PQ protocol will enable the QC/QA Labs to initiate the performance qualification after the IQ andOQ have been completed following the respective separate protocol. The guidelines are GMP compliant and follow EP, USP and JP regulatory guidelines.
應用
Rapid Sterility Testing Validation Protocol is used to validate rapid sterility testing procedures with ease and consistent performance while following regulatory guidelines.
法律資訊
MILLIFLEX is a registered trademark of Merck KGaA, Darmstadt, Germany
分析證明 (COA)
輸入產品批次/批號來搜索 分析證明 (COA)。在產品’s標籤上找到批次和批號,寫有 ‘Lot’或‘Batch’.。
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務