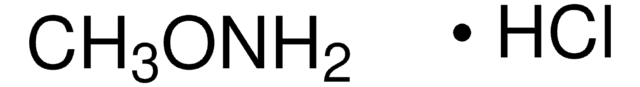262017
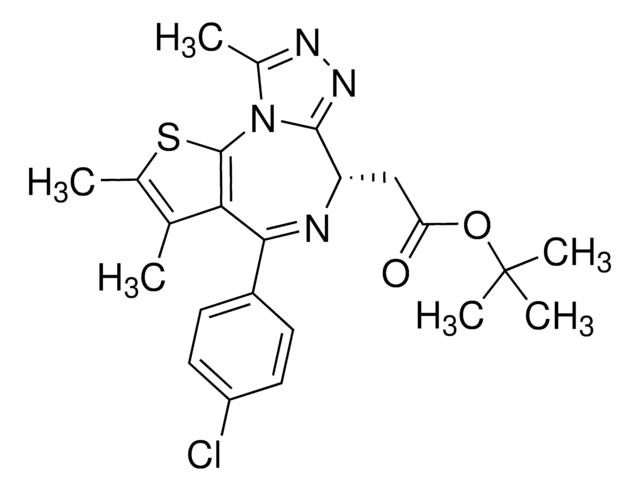
APE1 Inhibitor III
The APE1 Inhibitor III controls the biological activity of APE1. This small molecule/inhibitor is primarily used for Cell Structure applications.
同義詞:
APE1 Inhibitor III, Apurinic Endonuclease 1 Inhibitor III, Apurinic/Apyrimidinic Endonuclease 1 Inhibitor III, N-(3-(1,3-Benzo[d]thiazol-2-yl)-6-isopropyl-4,5,6,7-tetrahydrothieno[2,3-c]pyridin-2-yl)acetamide, N-(3-(1,3-Benzo[d]thiazol-2-yl)-6-isopropyl-4,5,6,7-tetrahydrothieno[2,3-c]pyridin-2-yl)acetamide, Apurinic Endonuclease 1 Inhibitor III, Apurinic/Apyrimidinic Endonuclease 1 Inhibitor III
About This Item
推薦產品
品質等級
化驗
≥95% (HPLC)
形狀
solid
製造商/商標名
Calbiochem®
儲存條件
OK to freeze
protect from light
顏色
brown
溶解度
DMSO: 2.5 mg/mL
運輸包裝
ambient
儲存溫度
2-8°C
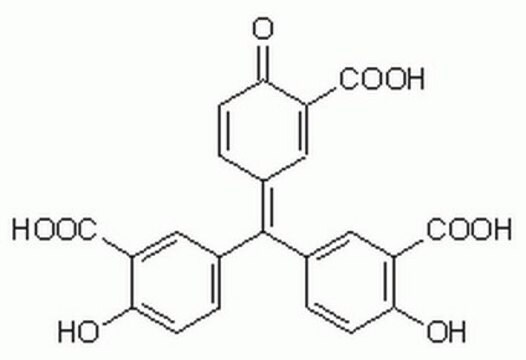
SMILES 字串
CC(C)N(CC1)CC2=C1C(C3=NC4=C(C=CC=C4)S3)=C(S2)NC(C)=O
InChI
1S/C19H21N3OS2/c1-11(2)22-9-8-13-16(10-22)25-18(20-12(3)23)17(13)19-21-14-6-4-5-7-15(14)24-19/h4-7,11H,8-10H2,1-3H3,(H,20,23)
InChI 密鑰
JMSPCTGDYFVMJZ-UHFFFAOYSA-N
一般說明
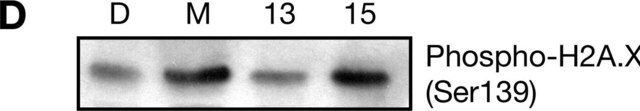
生化/生理作用
APE1
包裝
警告
重構
其他說明
法律資訊
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 2
閃點(°F)
Not applicable
閃點(°C)
Not applicable
分析證明 (COA)
輸入產品批次/批號來搜索 分析證明 (COA)。在產品’s標籤上找到批次和批號,寫有 ‘Lot’或‘Batch’.。
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務