推薦產品
品質等級
化驗
≥95%
形狀
powder
mp
230-234 °C
SMILES 字串
CC(C)(C)C1=CC([N+](C2=CC=C(C(F)(F)F)C=C2)=CO3)=C3C(C(C)(C)C)=C1.F[B-2](F)(F)F
應用
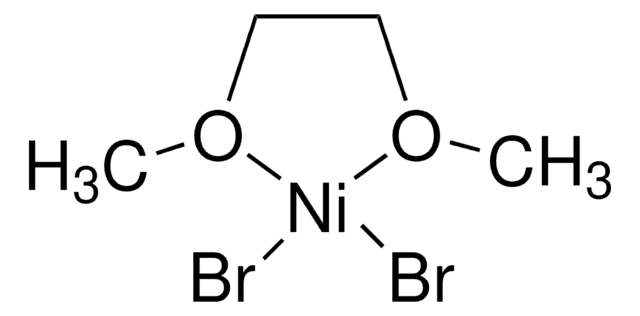
Deoxazole-quat is an N-heterocyclic carbene salt which activates tertiary free alcohols for a C-C cross coupling reaction with aryl halides. The MacMillan group developed a mild, robust, and selective metallaphotoredox-based cross-coupling platform for the deoxygenative coupling for free alcohols using Deoxazole-quat, a Ni catalyst, and [Ir(dtbbpy)(ppy)2]PF6.
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
Zhe Dong et al.
Nature, 598(7881), 451-456 (2021-09-01)
Metal-catalysed cross-couplings are a mainstay of organic synthesis and are widely used for the formation of C-C bonds, particularly in the production of unsaturated scaffolds1. However, alkyl cross-couplings using native sp3-hybridized functional groups such as alcohols remain relatively underdeveloped2. In particular
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務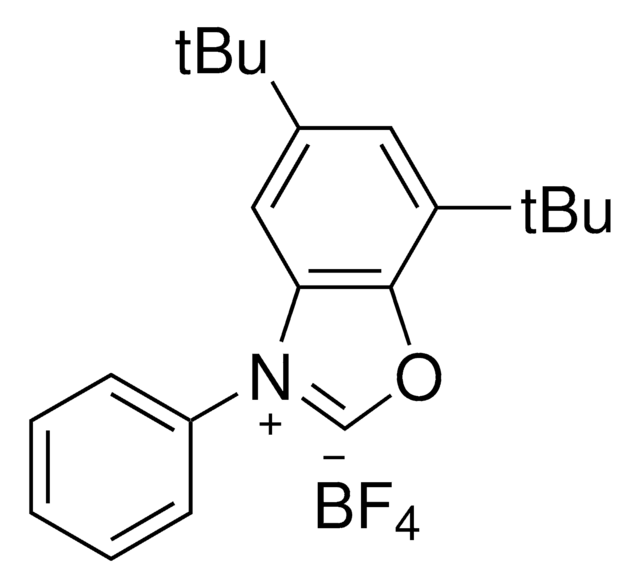
![(Ir[dF(CF3)ppy]2(dtbpy))PF6](/deepweb/assets/sigmaaldrich/product/structures/982/913/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09/640/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09.png)

![[Ir(dtbbpy)(ppy)2]PF6](/deepweb/assets/sigmaaldrich/product/structures/158/329/2544d673-d267-4aa1-8f46-2652aad4bfa0/640/2544d673-d267-4aa1-8f46-2652aad4bfa0.png)


![[铱 (dF (Me) ppy)] 2 (dtbbpy) ] PF 6](/deepweb/assets/sigmaaldrich/product/structures/150/099/7c2dfa31-39f4-4cca-aee5-86d4a89fea78/640/7c2dfa31-39f4-4cca-aee5-86d4a89fea78.png)