推薦產品
化驗
97% (HPLC)
形狀
powder or crystals
光學活性
[α]22/D -25.0°, c = 0.5% in dichloromethane
反應適用性
reaction type: Fmoc solid-phase peptide synthesis
mp
234-239 °C
應用
peptide synthesis
官能基
Fmoc
儲存溫度
2-8°C
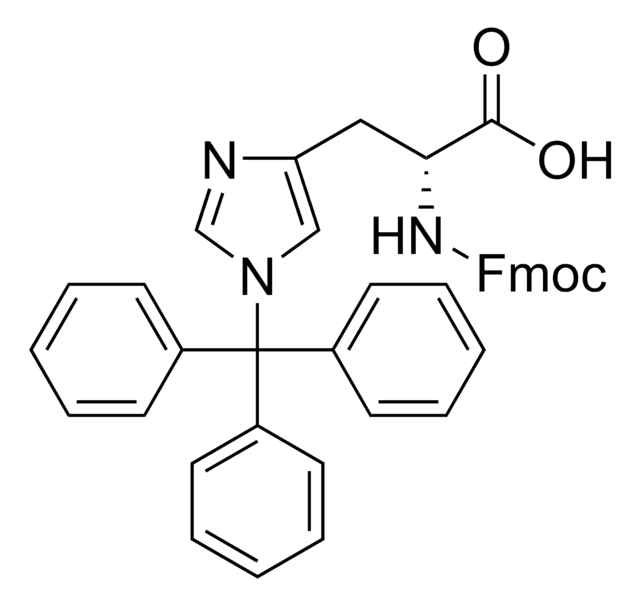
SMILES 字串
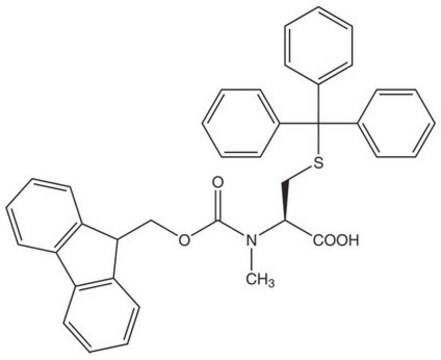
S(C[C@H](N(C)C(=O)OCC4c5c(cccc5)c6c4cccc6)C(=O)O)C(c3ccccc3)(c2ccccc2)c1ccccc1
InChI
1S/C38H33NO4S/c1-39(37(42)43-25-34-32-23-13-11-21-30(32)31-22-12-14-24-33(31)34)35(36(40)41)26-44-38(27-15-5-2-6-16-27,28-17-7-3-8-18-28)29-19-9-4-10-20-29/h2-24,34-35H,25-26H2,1H3,(H,40,41)/t35-/m0/s1
InChI 密鑰
RAKOPMQMPUNRGI-DHUJRADRSA-N
相關類別
應用
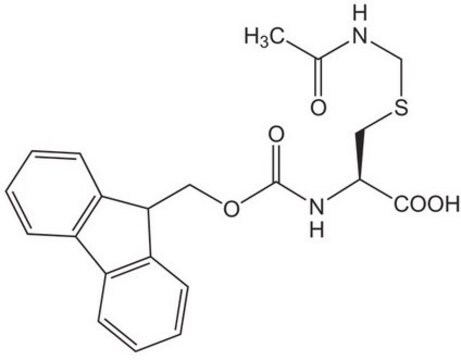
Fmoc-N-Me-Cys(Trt)-OH is a Fmoc-protected derivative of N-methyl cysteine used as a building block to prepare peptide thioesters under acidic conditions. The residue attached to the amino group of N-methylcysteine can migrate to the cysteinyl thiol group, resulting in the formation of a peptide thioester. The introduction of this Fmoc-protected derivative is best achieved using HATU as a coupling reagent in the presence of DIPEA (N, N-Diisopropylethylamine).
It can also be used to prepare Fmoc-N-Me-Cys(Trt)-OAllyl intermediate for the solid-phase synthesis of dithiol Triostin A.
It can also be used to prepare Fmoc-N-Me-Cys(Trt)-OAllyl intermediate for the solid-phase synthesis of dithiol Triostin A.
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務