推薦產品
品質等級
化驗
97%
形狀
solid
mp
233-237 °C
官能基
ether
fluoro
儲存溫度
2-8°C
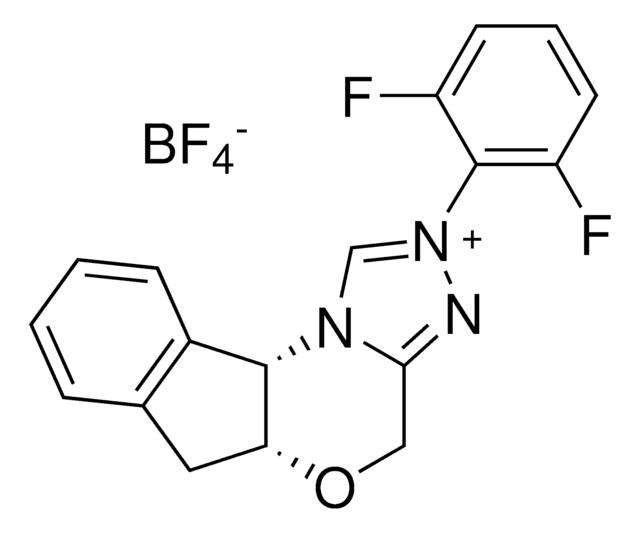
SMILES 字串
F[B-](F)(F)F.Fc1c(F)c(F)c(c(F)c1F)-[n+]2c[n@@H]3[C@@H]4[C@@H](Cc5ccccc45)OCc3n2
InChI
1S/C18H11F5N3O.BF4/c19-12-13(20)15(22)18(16(23)14(12)21)26-7-25-11(24-26)6-27-10-5-8-3-1-2-4-9(8)17(10)25;2-1(3,4)5/h1-4,7,10,17H,5-6H2;/q+1;-1/t10-,17+;/m1./s1
InChI 密鑰
CPCMDOOTVHDRTM-CVJFODCESA-N
應用
- Benzoin reactions of aldehydes and total synthesis of a natural product named isodarparvinol B.
- Synthesis of spirocyclic oxindole-dihydropyranones by reacting α-bromo-α,β-unsaturated aldehydes with isatin derivatives.
- Synthesis of 1,4-dicarbonyl compounds through intramolecular Stetter reaction.
訊號詞
Danger
危險聲明
危險分類
Eye Dam. 1 - Skin Corr. 1B
儲存類別代碼
8A - Combustible corrosive hazardous materials
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
文章
Rovis has demonstrated that triazolium salt in the presence of a base can act as an N-heterocyclic carbene organocatalyst in highly enantioselective intramolecular Stetter reactions.
Rovis has demonstrated that triazolium salt in the presence of a base can act as an N-heterocyclic carbene organocatalyst in highly enantioselective intramolecular Stetter reactions.
Global Trade Item Number
| 庫存單位 | GTIN |
|---|---|
| 674788-250MG | 4061833483732 |
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務![6,7-二氢-2-五氟苯基-5H-吡咯并[2,1-c]-1,2,4-三唑鎓四氟硼酸盐 97%](/deepweb/assets/sigmaaldrich/product/structures/338/297/58bedd1e-fa2f-4f2d-92eb-c61b56af8ded/640/58bedd1e-fa2f-4f2d-92eb-c61b56af8ded.png)
![(5aR,10bS)-5a,10b-二氢-2-(2,4,6-三甲苯基)-4H,6H-茚并[2,1-b]-1,2,4-三唑并[4,3-d]-1,4-噁嗪鎓氯化物 93%](/deepweb/assets/sigmaaldrich/product/structures/104/483/183b49bc-426f-411b-8d11-71bbd4b81022/640/183b49bc-426f-411b-8d11-71bbd4b81022.png)

![2-均三甲苯基-2,5,6,7-四氢吡咯并[2,1-c][1,2,4]三唑-4-鎓氯化物 97%](/deepweb/assets/sigmaaldrich/product/structures/267/516/0a2e9bce-0442-44c8-b912-3f3eeae583cf/640/0a2e9bce-0442-44c8-b912-3f3eeae583cf.png)
![(5aS,10bR)-5a,10b-Dihydro-2-mesityl-4H,6H-indeno[2,1-b]-1,2,4-triazolo[4,3-d]-1,4-oxazinium chloride](/deepweb/assets/sigmaaldrich/product/structures/226/284/a4e3161c-6ede-440f-8902-6af8e576a3ab/640/a4e3161c-6ede-440f-8902-6af8e576a3ab.png)
![(S)-5-苄基-2-均三甲苯基-6,6-二甲基-6,8-二氢-5H-[1,2,4]三氮唑并[3,4-c][1,4]噁嗪-2-四氟硼酸鎓盐](/deepweb/assets/sigmaaldrich/product/structures/264/074/1f7b927e-bdc3-4352-9a96-ff81396c1618/640/1f7b927e-bdc3-4352-9a96-ff81396c1618.png)
![(5R,6S)-2-Mesityl-5,6-diphenyl-6,8-dihydro-5H-[1,2,4]triazolo[3,4-c][1,4]oxazin-2-ium tetrafluoroborate 97%](/deepweb/assets/sigmaaldrich/product/structures/219/182/9bfa803e-8970-4dd1-9cbf-9ebca2f74da2/640/9bfa803e-8970-4dd1-9cbf-9ebca2f74da2.png)