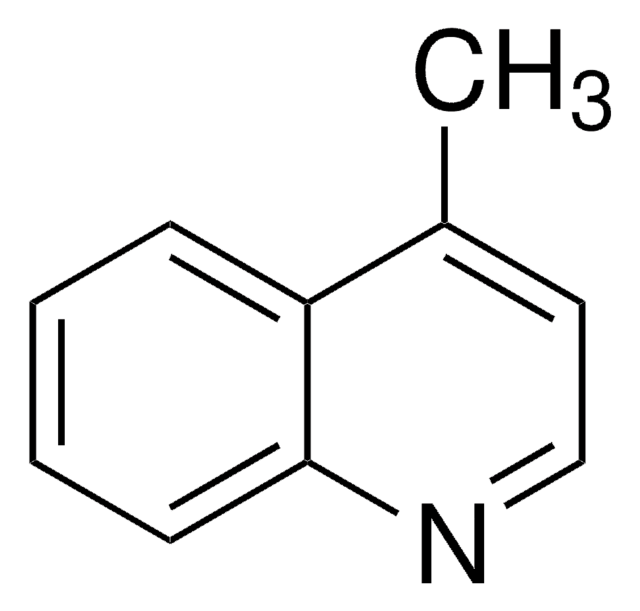
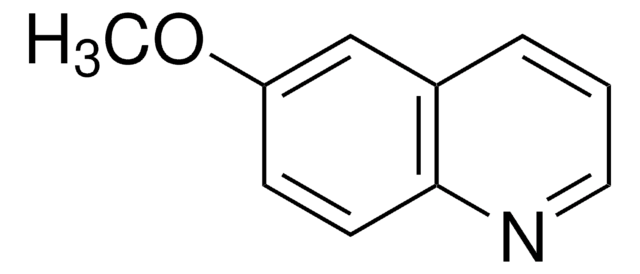
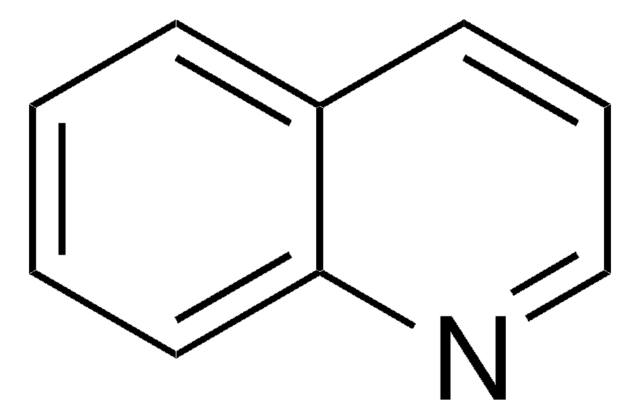
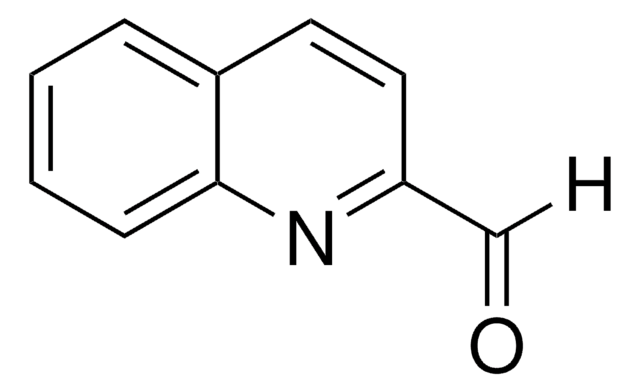
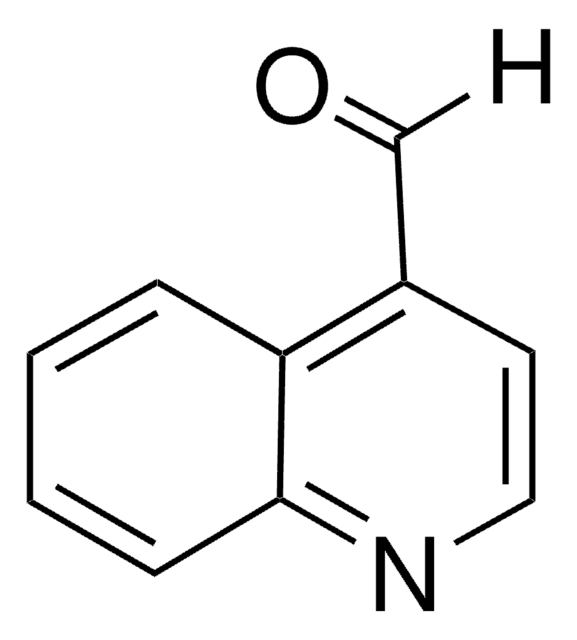
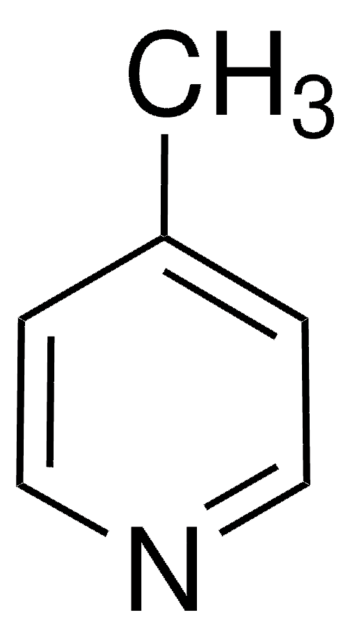
推薦產品
化驗
99%
形狀
liquid
折射率
n20/D 1.615 (lit.)
bp
252-253 °C (lit.)
mp
16-17 °C (lit.)
密度
1.069 g/mL at 25 °C (lit.)
SMILES 字串
Cc1cnc2ccccc2c1
InChI
1S/C10H9N/c1-8-6-9-4-2-3-5-10(9)11-7-8/h2-7H,1H3
InChI 密鑰
DTBDAFLSBDGPEA-UHFFFAOYSA-N
基因資訊
human ... CYP1A2(1544)
一般說明
3-Methylquinoline is a quinoline derivative. It is widely employed for the synthesis of dyes, food coloring agents, pharmaceutical reagents, pH indicators and in various industrial processes. It has been synthesized by the methylation of quinoline with methanol in the presence of various zeolites in a fixed-bed reactor.
應用
3-Methylquinoline may be used as carbon, nitrogen and energy supplement to investigate its degradation by Comamonas testosteroni 63. It may be used as ligand in the preparation of tetra-μ-benzoato-bis[(3-methylquinoline)copper(II)], a paddle-wheel-type dinuclear complex.
訊號詞
Danger
危險分類
Acute Tox. 4 Dermal - Carc. 2 - Eye Dam. 1 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
10 - Combustible liquids
水污染物質分類(WGK)
WGK 3
閃點(°F)
235.4 °F - closed cup
閃點(°C)
113 °C - closed cup
個人防護裝備
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
客戶也查看了
Byeong Kwon Park et al.
Acta crystallographica. Section E, Structure reports online, 64(Pt 9), m1141-m1141 (2008-01-01)
In the title compound, [Cu(2)(C(7)H(5)O(2))(4)(C(10)H(9)N)(2)], the paddle-wheel-type dinuclear complex mol-ecule contains four bridging benzoate groups and two terminal 3-methyl-quinoline ligands. The asymmetric unit contains one and a half mol-ecules with a total of three independent Cu atoms; there is an
Selective synthesis of 2-methylquinoline over zeolites.
Reddy PR, et al.
Catalysis Letters, 56(2-3), 155-158 (1998)
S Schach et al.
Biological chemistry Hoppe-Seyler, 374(3), 175-181 (1993-03-01)
A bacterial strain which utilizes 3-methylquinoline as sole source of carbon, nitrogen and energy was isolated from activated sludge. On the basis of its morphological and physiological characteristics, this isolate was classified as Comamonas testosteroni. Four metabolites of 3-methylquinoline degradation
S Schach et al.
European journal of biochemistry, 232(2), 536-544 (1995-09-01)
The enzymes catalysing the first two steps of quinoline and 3-methylquinoline degradation by Comamonas testosteroni 63 were investigated. Quinoline 2-oxidoreductase, which catalyses the hydroxylation of (3-methyl-)quinoline to (3-methyl-)2-oxo-1,2-dihydroquinoline, was purified to apparent homogeneity. The native enzyme, with a molecular mass
Tamara Rodríguez-Cabo et al.
Analytical and bioanalytical chemistry, 407(20), 6159-6170 (2015-06-05)
Time-of-flight accurate mass spectrometry (TOF-MS), following a previous chromatographic (gas or liquid chromatography) separation step, is applied to the identification and structural elucidation of quinoline-like alkaloids in honey. Both electron ionization (EI) MS and positive electrospray (ESI+) MS spectra afforded
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務