推薦產品
化驗
99%
形狀
solid
bp
250-260 °C (lit.)
mp
93-96 °C (lit.)
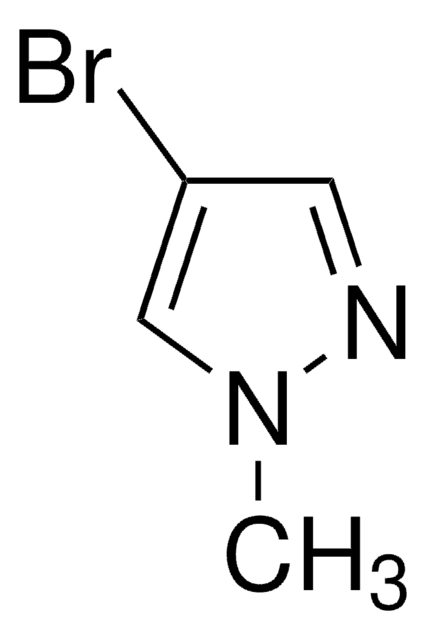
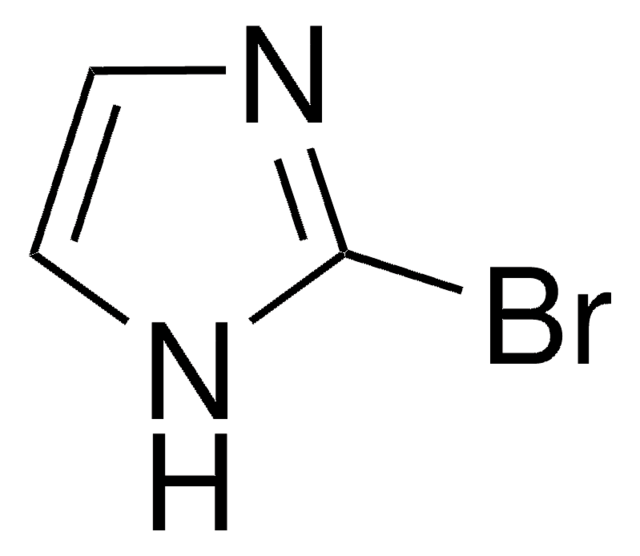
官能基
bromo
SMILES 字串
Brc1cn[nH]c1
InChI
1S/C3H3BrN2/c4-3-1-5-6-2-3/h1-2H,(H,5,6)
InChI 密鑰
WVGCPEDBFHEHEZ-UHFFFAOYSA-N
一般說明
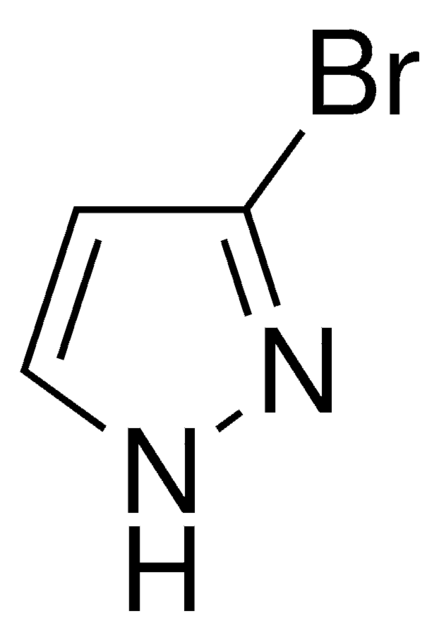
4-溴吡唑是一种吡唑衍生物。它被报道可与四氯化钛反应生成二元加合物。4-溴吡唑的致突变性已通过使用鼠伤寒沙门氏菌的L-阿拉伯糖正向突变分析进行了检测。据报道,它可抑制氧化磷酸化、ATP-32P交换反应以及能量依赖型和非依赖型的钙摄取。
4-溴吡唑是一种杂芳基卤化物,它在钯催化剂存在下的氰化已被报道。
應用
4-溴吡唑可用于制备4-溴-1-(2-氯乙基)-1H-吡唑。它可作为合成1,4′-双吡唑的起始材料。
通过与二甲基和二乙烯基锡二氯化物反应,4-溴吡唑可用于制备固态的六配位复合物。
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
客戶也查看了
E Alejandre-Durán et al.
Environmental mutagenesis, 8(4), 611-619 (1986-01-01)
The mutagenicity of pyrazole and seven pyrazole derivatives (4-nitropyrazole, 4-bromopyrazole, 1-methyl-4-nitropyrazole, 3,5-dimethyl-4-nitropyrazole, 1-methyl-4-bromopyrazole, 4,4'-dinitro-1, 1'-methylene-dipyrazole and 4,4'-dibromo-1,1'-methylene-dipyrazole) has been investigated with the L-arabinose forward mutation assay of Salmonella typhimurium. Two nitroimidazoles (1-methyl-5-nitroimidazole and metronidazole) were included as reference drugs. The
Inhibition of the oxidation of the urinary bladder carcinogen N-butyl-N-(4-hydroxybutyl)nitrosamine by pyrazole and 4-substituted pyrazoles.
C C Irving et al.
Biochemical pharmacology, 37(8), 1642-1644 (1988-04-15)
Tetrahedron, 63, 748-748 (2007)
Dichlorodialkyltin complexes with 4-bromopyrazole. The crystal structure of bis(4-bromopyrazole- N2)dichlorodimethyltin(IV).
Casellato U, et al.
Journal of Organometallic Chemistry, 486(1-2), 105-107 (1995)
Ilia A. Guzei et al.
Inorganic chemistry, 36(20), 4415-4420 (2001-10-24)
Treatment of titanium tetrachloride with 3,5-di-tert-butylpyrazole affords the complexes [3,5-(C(CH(3))(3))(2)C(3)H(3)N(2)](2)[TiCl(6)] and (3,5-(C(CH(3))(3))(2)C(3)HN(2))(2)TiCl(2) in 37 and 42% yields, respectively. An analogous reaction with 3,5-dimethylpyrazole, 3-methylpyrazole, 4-bromopyrazole, and 4-iodopyrazole leads to the formation of corresponding TiCl(4)L(2) binary adducts in 30-86% yields. Crystal
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務