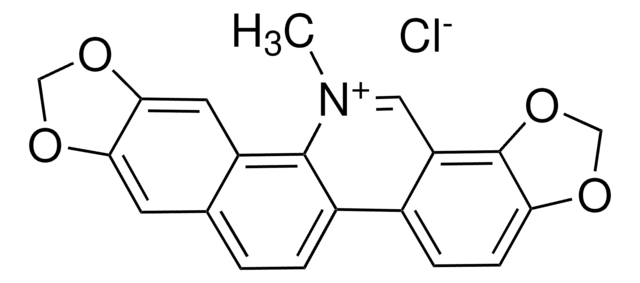
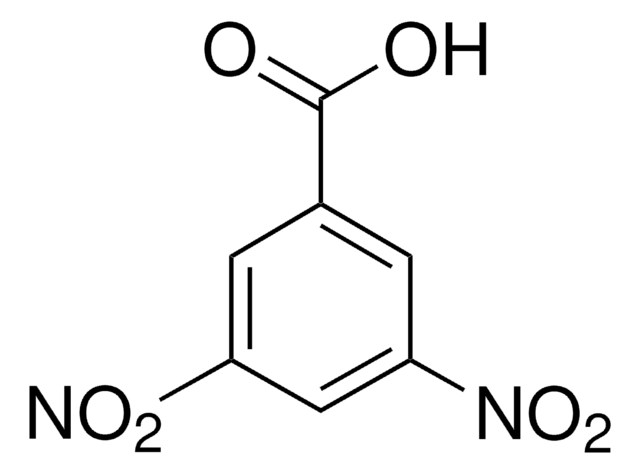
推薦產品
品質等級
化驗
97%
雜質
~1.5 mol/mol methanol
mp
206-207 °C (dec.) (lit.)
SMILES 字串
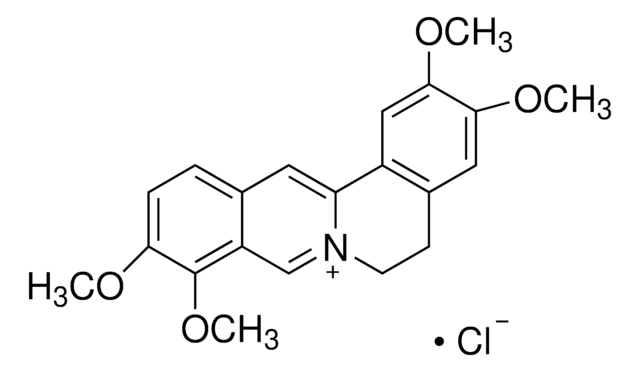
[Cl-].[H]O[H].COc1cc2CC[n+]3cc4c(OC)c(OC)ccc4cc3-c2cc1OC
InChI
1S/C21H22NO4.ClH.H2O/c1-23-18-6-5-13-9-17-15-11-20(25-3)19(24-2)10-14(15)7-8-22(17)12-16(13)21(18)26-4;;/h5-6,9-12H,7-8H2,1-4H3;1H;1H2/q+1;;/p-1
InChI 密鑰
PIQNSCSNSSZUIT-UHFFFAOYSA-M
一般說明
Palmatine chloride hydrate (Palmatine) is an alkaloid. It is a potential phototoxin, and exhibits low quantum yields for fluorescence. Normal Raman spectra and DFT calculations of palmatine chloride hydrate is reported.
應用
Palmatine chloride hydrate (Palmatine) is suitable for use:
- as alkaloid standard in the method validation for determination of berberine, hydrastine and canadine in goldenseal (Hydrastis canadensis L.) root powder
- in the preparation of 8-heteroaryl-7,8-dihydroprotoberberine
- in a study to investigate the FT-Raman and surface-enhanced Raman scattering (SERS) spectra of three related alkaloid dyes, namely palmatine, jatrorrhizine and coptisine
訊號詞
Warning
危險分類
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
235.4 °F - closed cup
閃點(°C)
113 °C - closed cup
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
客戶也查看了
Fluorescence enhancement for alkaloids by anions: Spectroscopic and electrochemical characterizations.
Chung Y-L, et al.
Journal of Electroanalytical Chemistry, 610(1), 85-89 (2007)
Lenka Grycová et al.
Magnetic resonance in chemistry : MRC, 46(12), 1127-1134 (2008-09-11)
Adducts of the quaternary protoberberine alkaloids (QPA) berberine, palmatine, and coptisine were prepared with nucleophiles derived from pyrrole, pyrazole, imidazole, and 1,2,4-triazole. The products, 8-substituted 7,8-dihydroprotoberberines, were identified by mass spectrometry and 1D and 2D NMR spectroscopy, including (1)H--(15)N shift
Sayaka Shinji et al.
Bioscience, biotechnology, and biochemistry, 84(1), 63-75 (2019-08-30)
A natural isoquinoline alkaloid, berberine, has been known to exhibit anti-tumor activity in various cancer cells via inducing cell cycle arrest. However, it has not been investigated whether berberine and its analogs inhibit the growth of rhabdomyosarcoma (RMS), which is
Surface-enhanced Raman scattering of protoberberine alkaloids.
Canamares MV, et al.
Journal of Raman Spectroscopy, 39(12), 1907-1914 (2008)
Holly A Weber et al.
Journal of AOAC International, 86(3), 476-483 (2003-07-11)
A fast, practical ambient extraction methodology followed by isocratic liquid chromatography (LC) analysis with UV detection was validated for the determination of berberine, hydrastine, and canadine in goldenseal (Hydrastis canadensis L.) root powder. The method was also validated for palmatine
Global Trade Item Number
| 庫存單位 | GTIN |
|---|---|
| 361615-100MG | 4061831824339 |
| 361615-500MG | 4061831824346 |
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務