265004
(4R,5R)-2,2-二甲基-α,α,α′,α′-四苯基-1,3-二氧戊环-4,5-二甲醇
97%
同義詞:
(-)-2,3-O-异亚丙基-1,1,4,4-四苯基-L-苏糖醇, (-)-反式-α,α′-(2,2-二甲基-1,3-二氧戊环-4,5-二基)双(二苯基甲醇), (4R,5R)-4,5-双(二苯基羟甲基)-2,2-二甲基二氧戊环, 1,1,4,4-四苯基-2,3-O-异亚丙基-L-苏糖醇, TADDOL
About This Item
推薦產品
化驗
97%
光學活性
[α]19/D −62.6°, c = 1 in chloroform
mp
193-195 °C (lit.)
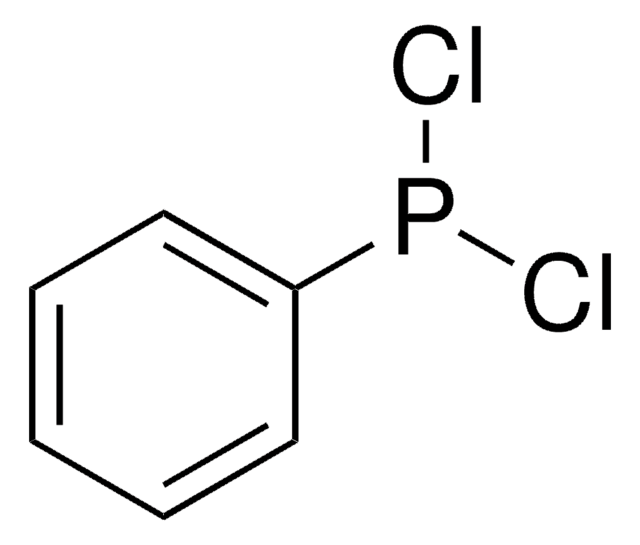
SMILES 字串
CC1(C)O[C@H]([C@@H](O1)C(O)(c2ccccc2)c3ccccc3)C(O)(c4ccccc4)c5ccccc5
InChI
1S/C31H30O4/c1-29(2)34-27(30(32,23-15-7-3-8-16-23)24-17-9-4-10-18-24)28(35-29)31(33,25-19-11-5-12-20-25)26-21-13-6-14-22-26/h3-22,27-28,32-33H,1-2H3/t27-,28-/m1/s1
InChI 密鑰
OWVIRVJQDVCGQX-VSGBNLITSA-N
應用
Catalyst involved in synthesis of cyclopropylamines via addition reactions of Grignard reagents to amides
Reactant or reagent involved in:
- Enantioswitching of catalytic asymmetric hydroboration
- Synthesis of derivative ligands for asymmetric hydroformylation of alkenes
- Amide-directed catalytic asymmetric hydroboration of trisubstituted alkenes
- Addition of deactivated alkyl Grignard reagents to aldehydes
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Gloves, type N95 (US)
分析證明 (COA)
輸入產品批次/批號來搜索 分析證明 (COA)。在產品’s標籤上找到批次和批號,寫有 ‘Lot’或‘Batch’.。
客戶也查看了
文章
The chiral auxiliaries TADDOLs (α,α,α,α-tetraaryl-1,3-dioxolane-4,5- dimethanols) developed by Seebach's group have found numerous applications in asymmetric synthesis ranging from utilization as stoichiometric chiral reagents or in Lewis acid mediated reactions, to roles in catalytic hydrogenation and stereoregular metathesis polymerization.
Apart from numerous examples using TADDOLs in metal-catalyzed asymmetric reactions, Rawal recently reported that TADDOLs could be used as Brønsted acid organocatalysts in highly stereoselective hetero-Diels–Alder reactions.
Chiral Diols
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務

![[Ir(dFCF3ppy)2-(5,5’-dCF3bpy)]PF6 ≥95%](/deepweb/assets/sigmaaldrich/product/structures/422/901/e00f3148-fb86-4f94-9e79-21d064c3f327/640/e00f3148-fb86-4f94-9e79-21d064c3f327.png)