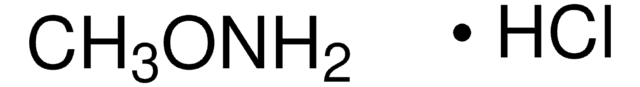全部照片(3)
About This Item
線性公式:
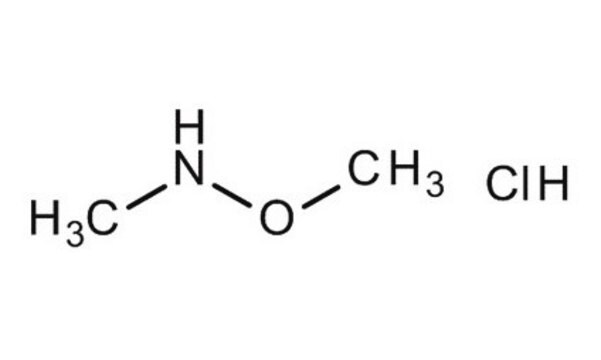
(CH3)2NOH · HCl
CAS號碼:
分子量::
97.54
Beilstein:
3905683
EC號碼:
MDL號碼:
分類程式碼代碼:
12352100
PubChem物質ID:
NACRES:
NA.22
推薦產品
化驗
99%
形狀
solid
mp
107-109 °C (lit.)
SMILES 字串
Cl[H].CN(C)O
InChI
1S/C2H7NO.ClH/c1-3(2)4;/h4H,1-2H3;1H
InChI 密鑰
HWWVAHCWJLGKLW-UHFFFAOYSA-N
尋找類似的產品? 前往 產品比較指南
應用
N,N-二甲基羟胺盐酸盐用于合成 4,4-二甲基-2,5,5-三苯基-1.3-二氧杂-4-氮杂-2-硼-2-硼酸环戊烷。boratacyclopentane它也被用作聚合物链终止剂。
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
分析證明 (COA)
輸入產品批次/批號來搜索 分析證明 (COA)。在產品’s標籤上找到批次和批號,寫有 ‘Lot’或‘Batch’.。
客戶也查看了
Structural studies of organoboron compounds. XVI. Preparation and crystal and molecular structures of 4, 4-dimethyl-2, 5, 5-triphenyl-1, 3-dioxa-4-azonia-2-bora-5-boratacyclopentane and 4, 4, 5, 5-tetramethyl-2, 2-diphenyl-1, 3-dioxa-4-azonia-2-boratacyclopentane.
Canadian Journal of Chemistry, 62(5), 838-844 (1984)
Hana Popelkova et al.
Photosynthesis research, 110(2), 111-121 (2011-11-02)
The photosystem II (PSII) manganese-stabilizing protein (PsbO) is known to be the essential PSII extrinsic subunit for stabilization and retention of the Mn and Cl(-) cofactors in the oxygen evolving complex (OEC) of PSII, but its function relative to Ca(2+)
A R Tunoori et al.
Organic letters, 2(25), 4091-4093 (2000-12-12)
[structure] The reagent [bis(2-methoxyethyl)amino]sulfur trifluoride (Deoxo-Fluor reagent) converts carboxylic acids to the corresponding acid fluorides, which then react with N,N-dimethylhydroxylamine to give the corresponding Weinreb amides in high yields. The reaction proceeds without racemization when optically active acids are used
K Stolze et al.
Free radical research communications, 8(2), 123-131 (1990-01-01)
Nitroxide radicals have been detected in the methemoglobin formation reaction between oxyhemoglobin and the substituted hydroxylamine compounds, N-methylhydroxylamine and N,N-dimethylhydroxylamine, by ESR spectroscopy. The stability of these nitroxide radicals was considerably higher than that of the NH2O. radical derived from
J Taira et al.
Biochimica et biophysica acta, 1336(3), 502-508 (1997-11-21)
Hydroxylamine (HA), which is a natural product of mammalian cells, has been shown to possess vasodilatory properties in several model systems. In this study, HA and methyl-substituted hydroxylamines, N-methylhydroxylamine (NMHA) and N,N-dimethylhydroxylamine (NDMHA), have been tested for their ability to
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務