推薦產品
品質等級
化驗
95%
折射率
n20/D 1.474 (lit.)
bp
58 °C/13 mmHg (lit.)
密度
0.87 g/mL at 25 °C (lit.)
SMILES 字串
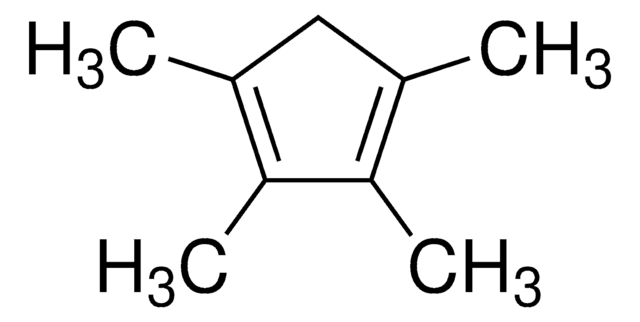
CC1C(C)=C(C)C(C)=C1C
InChI
1S/C10H16/c1-6-7(2)9(4)10(5)8(6)3/h6H,1-5H3
InChI 密鑰
WQIQNKQYEUMPBM-UHFFFAOYSA-N
尋找類似的產品? 前往 產品比較指南
一般說明
研究了顺磁性内层金属富勒烯与 1,2,3,4,5-五甲基环戊二烯的 Diels-Alder 反应机理。报道了 1,2,3,4,5-五甲基环戊二烯的三步法大规模合成。用激光烧蚀质谱法研究了 1,2,3,4,5-五甲基环戊二烯 (HCp*) 在气相中与钯离子的反应。它与富含电子的烯烃发生自由基阳离子催化的环加成反应,生成 Diels-Alder 产物。
應用
1,2,3,4,5-五甲基环戊二烯用作:
- 五羰基铁中的金属有机溶剂蒸汽沉积中的生长调节剂化学品。
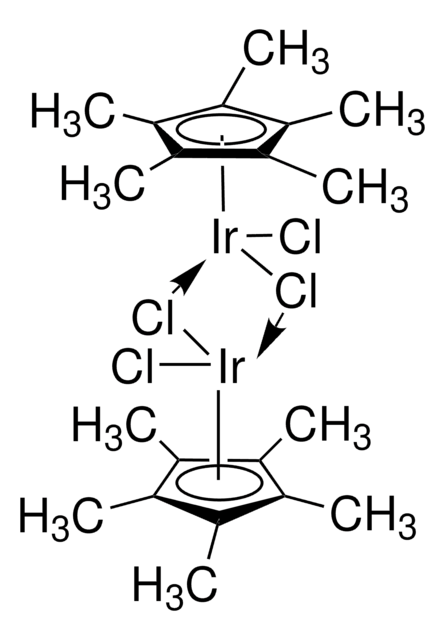
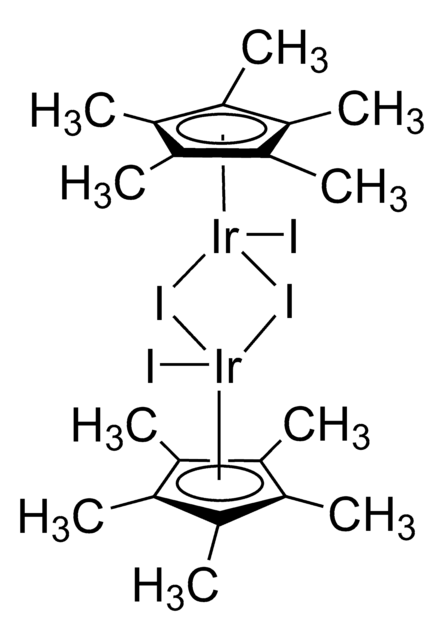
- 通过肟的中间体将醇催化转化为酰胺的“一锅法”铱中的配体。
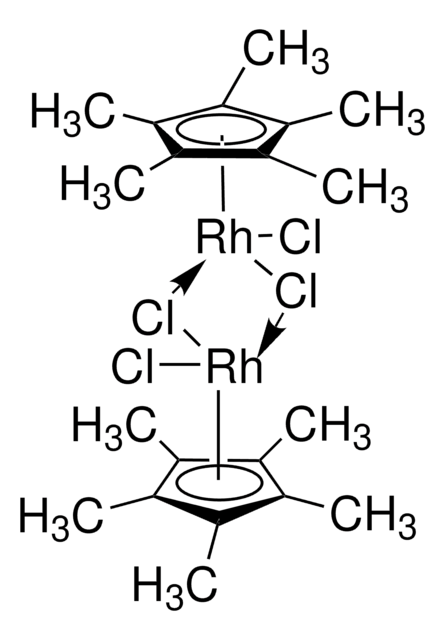
- 合成[Cp*Rh(bpy)H2O]2+(Cp* = 五甲基环戊二烯基,bpy = 2,2′-联吡啶),它是 NADH 再生过程中的电子介质。
客戶也查看了
Satoru Sato et al.
Journal of the American Chemical Society, 135(15), 5582-5587 (2013-03-23)
The reaction mechanism of the Diels-Alder reaction of paramagneticendohedral metallofullerene, La@C82, and 1,2,3,4,5-pentamethylcyclopentadiene was studied theoretically and experimentally. Theoretical calculations revealed that this reaction proceeds via a concerted mechanism that includes formation of a stable intermediate. The activation energy of
Reactions of actinide ions with pentamethylcyclopentadiene: atypical hydrocarbon activation.
Gibson JK.
International Journal of Mass Spectrometry, 202(1), 19-29 (2000)
Nathan A Owston et al.
Organic letters, 9(1), 73-75 (2006-12-29)
[reaction: see text] The iridium catalyst [Ir(Cp*)Cl2]2 is effective for the rearrangement of oximes to furnish amides. The reaction has been combined with catalytic transfer hydrogenation between an alcohol and alkene to allow the conversion of alcohols into amides in
The Aminium Salt and Photoinduced Electron Transfer Initiated Diels-Alder Cycloaddition of Electron-rich Allenes: Evidence for a Stepwise Mechanism and the Importance of Steric and Electronic Effects for the Reactivity of Distonic Radical Cation Intermediates.
Schmittel M, et al.
Chemistry (Weinheim An Der Bergstrasse, Germany), 2(8), 1031-1040 (1996)
Journal of Organometallic Chemistry, 472, 359-359 (1994)
文章
The Diels–Alder reaction is the reaction between a conjugated diene and an alkene (dienophile) to form unsaturated six-membered rings. It is also referred to as a cycloaddition.
The Diels–Alder reaction is the reaction between a conjugated diene and an alkene (dienophile) to form unsaturated six-membered rings. Since the reaction involves the formation of a cyclic product via a cyclic transition state, it is also referred to as a "cycloaddition".
Global Trade Item Number
| 庫存單位 | GTIN |
|---|---|
| 214027-25G | 4061838773081 |
| 214027-5G | 4061838773098 |
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務